Beneath the Surface: Perusing the Protein Powerhouse of Skincare
by Simon Currie, Ph.D.

by Simon Currie, Ph.D.
Do you know what our largest organ is? No, not the intestines, stomach, liver, nor brain. It’s our skin! Skin covers almost all of our body and plays an important barrier role in keeping viruses and bacteria out of us and our internal organs in.
But what exactly is skin made of? Like all of our other cells, skin is made of proteins, lipids, and nucleic acids. However, one protein in particular is super abundant in skin and helps it function properly: collagen (Figure 1). In fact, collagen makes up nearly a third of the total protein content in our bodies (Deshmukh et al., 2016)!
Numerous cosmetic and aesthetic treatments use proteins and peptides to keep our skin healthy and to provide a youthful appearance. Many of these applications work by ramping up the production of collagen in our skin cells, or by adjusting the interactions between skin cells and our nervous and immune systems.
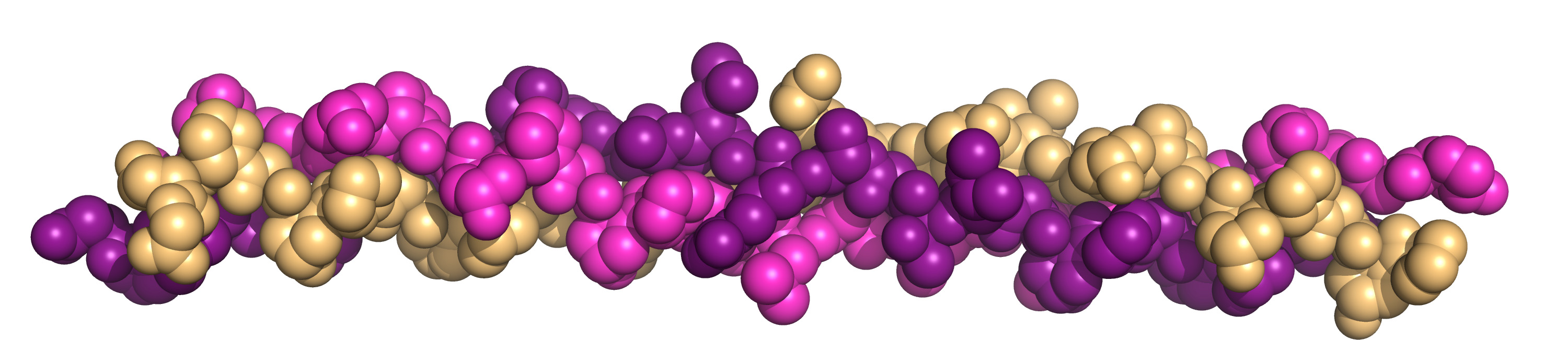
Figure 1. Collagen triple helix. Different colors
indicate distinct collagen strands (PDB: 1BKV).
You have likely seen ads for skincare products and lotions that promise to prevent aging and keep your skin looking young and healthy. Maybe you use these lotions yourself. Have you ever wondered or investigated what these concoctions contain?
The active ingredients in many cosmetics include proteins such as growth factors and peptides.
Growth factors are proteins that bind to the outside of cells to convey specific information to these cells. In the context of cosmetics, growth factors bind to skin cells to promote:
Your body naturally uses growth factors to maintain skin health. However, growth factors often dwindle as we age, so providing your skin with an extra boost of growth factors can enhance these processes.
Peptides are used in a number of cosmetics including anti-aging skin products. These peptides fall into distinct functional classes that:
For example, palmitoyl pentapeptide inhibits neurotransmitters, copper tripeptide inhibits inflammation, and acetyl hexapeptide stimulates the production of collagen (Figure 2).
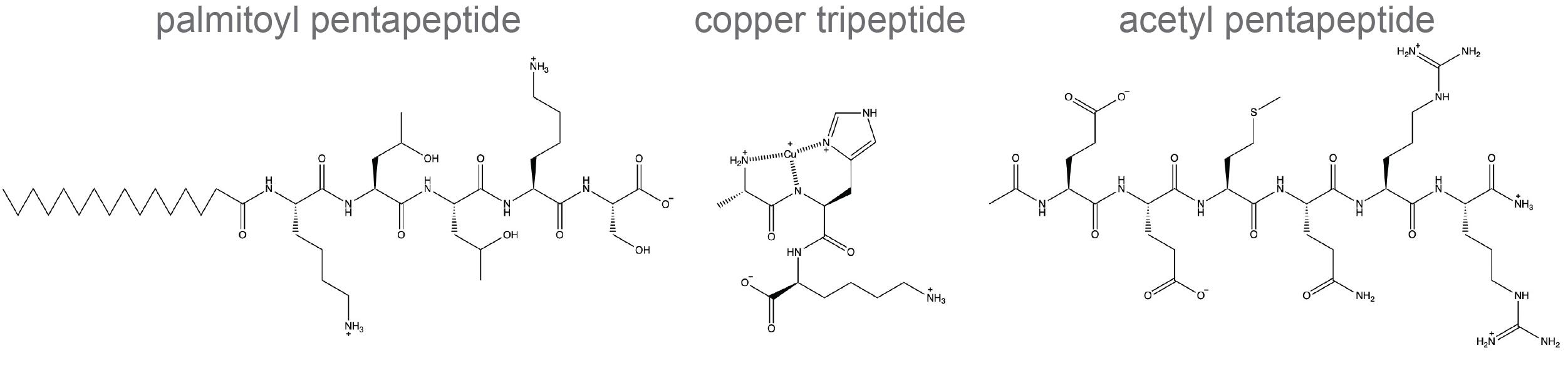
Figure 2. Examples
of cosmetic peptides.
Cosmetics usually contain multiple distinct peptides from each of these classes, resulting in mixtures of tens to hundreds of different peptides.
If you are up to date on the red carpet gossip, you probably know which movie stars are the latest to get Botox® or collagen fillers. Both of these treatments are examples of aesthetic dermatology.
Aesthetic skin treatments are intended to enhance skin appearance outside of the context of any kind of skin disease.
Botox® is a popular aesthetic treatment for aging and wrinkling skin. The active ingredient is a protein from the pathogen Clostridium botulinum, which causes botulism. Botulism attacks nerve cells and causes difficulty breathing, muscle paralysis, and even death if not treated properly.
Low doses of one C. botulinum neurotoxin, onabotulineurotoxin A (Figure 3), are locally injected in Botox® treatments to temporarily paralyze nerves within face muscles. Through this mechanism Botox® reduces wrinkles. However, new toxin must be reinjected every few months to maintain the wrinkle-free appearance due to its temporary nature.
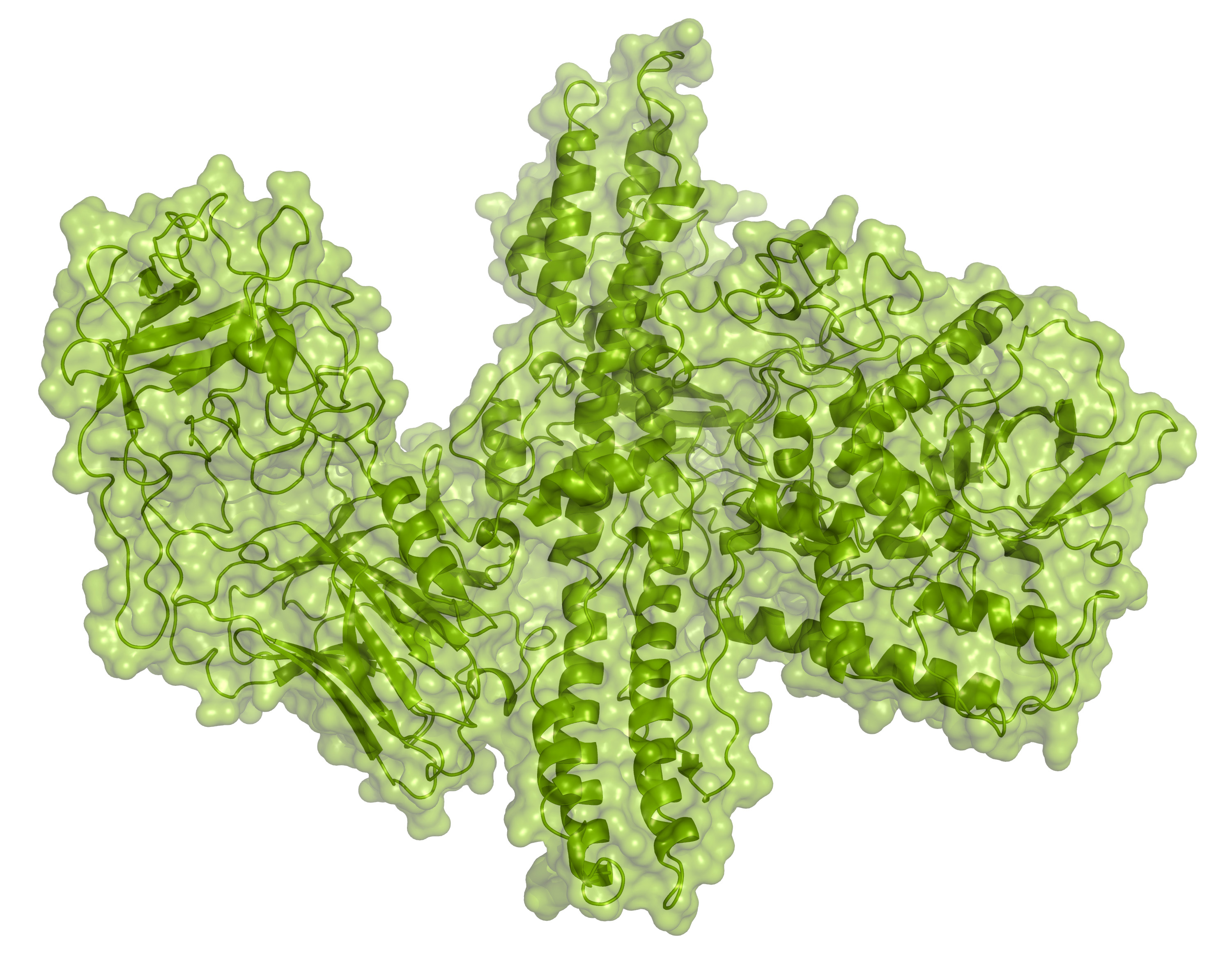
Figure 3.
Onabotulineurotoxin A, the active neurotoxin in Botox® (PDB: 3BTA).
Although most commonly known for its aesthetic applications, Botox® was originally developed as a medical treatment and is still used for a number of diseases involving spastic or misaligned muscles including:
Onabotulineurotoxin A is purified from C. botulinum cultures by dialysis and multiple acid/salt precipitations (Lamanna et al., 1946; Allergan Pharmaceuticals Inc., 2004). Dialysis separates onabotulineurotoxin A from smaller proteins and small organic molecules such as metabolites and nucleotides.
Most proteins precipitate in acidic pH and high concentrations of salts. Yet the pH, type of salt, and concentration of salt at which individual proteins precipitate varies. These unique properties allow scientists to perform a series of intentional protein precipitations to purify their protein of interest away from other cellular proteins.
This precipitation technique is used to purify onabuotulineurotoxin A away from other C. botulinum secreted proteins by performing a series of acid and salt precipitations using a combination of different pH values, and different types and concentrations of salts (Lamanna et al., 1946).
Since Botox® is a toxin, you may be wondering how it is dosed to give the desired effects on skin without accidentally poisoning people. Each batch of Botox® purified as described above is tested and given an activity “unit,” where the unit corresponds to the median lethal dose of that batch when injected into mice. For this reason, Botox® from different manufacturers, and even different batches from the same manufacturer, are not exchangeable.
Since we are much larger than mice, anywhere from 2 to 50 units of Botox® are injected, with the exact amount dependent on the nature of the injection site. For instance, 2 to 6 units are injected into the chin whereas 25-50 units are injected into the neck (Green, 2014).
Proper injection by a skilled healthcare professional minimizes the occurrence of side effects, which are usually mild and localized to the injection site.
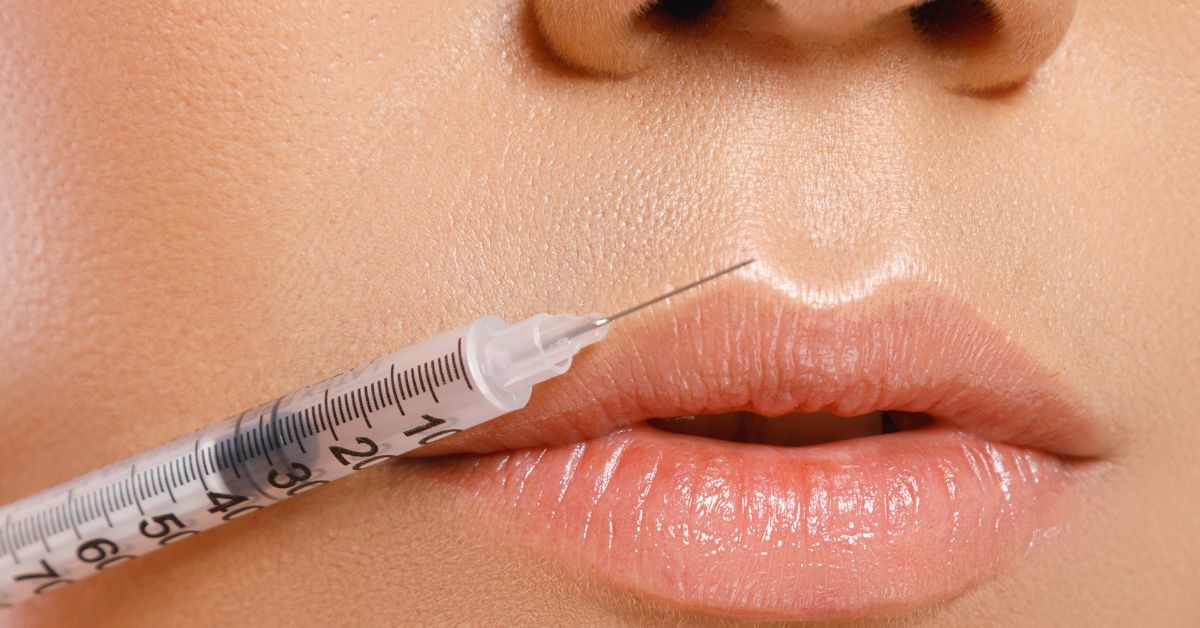
Figure 4. Collagen injections are thought to be more
effective and deeper penetrating than creams.
Collagen injections, also known as fillers, are another aesthetic skin treatment thought to give skin a fuller and more youthful look. Exogenous collagen can be injected deeper than surface creams reach, which some feel gives skin a plumper appearance.
The collagen used in fillers is purified from animal skins such as cowhide. Traditionally, harsh acid or base treatments were used to release collagen from the cowhide. However, researchers later discovered that proteases could cut collagen off of the cowhide instead of the acid and base treatments. This alternative process is more environmentally friendly as it eliminates toxic waste generation.
Collagen is the most abundant protein in these extractions and is additionally purified by:
Ion-exchange chromatography uses the intrinsic charge of a specific protein, collagen in this case, to purify it away from other cellular proteins.
Since animal collagen is used in fillers, a substantial minority of patients develop allergic reactions. An allergy test is performed on a small section of the forearm to try to uncover patients that develop allergic reactions before they develop a major rash on their face.
Over time, the proteases within our bodies will naturally degrade injected collagen. So, the fillers described above are a temporary fix and need to be updated on a monthly to yearly basis to maintain the desired appearance.
However, a recent advance on fillers uses cow collagen in combination with polymethylmethacrylate (PMMA) beads. After our proteases cleave the cow collagen, the inert PMMA beads remain, forming a scaffold that stimulates collagen production and infill from the surrounding skin cells. This longer-lasting formula persists for roughly 5 years and is the most common collagen injection used in the United States (Cockerham and Hsu, 2009).
Let’s wrap up this article on skin care. Hopefully you now know a little more about the powerful proteins that play key roles in cosmetic and aesthetic skin care applications, and how they are purified.
Allergan Pharmaceuticals Inc. (2004). Botox® [package insert]. Irvine, CA: Allergan Pharmaceuticals Inc.
Barañano, D. E., & Miller, N. R. (2004). Long term efficacy and safety of botulinum toxin A injection for crocodile tears syndrome. The British journal of ophthalmology, 88(4), 588–589. https://doi.org/10.1136/bjo.2003.029181
Beck, L. A., Thaci, D., Hamilton, J. D., Graham, N. M., Bieber, T., Rocklin, R., . . . Radin, A. R. (2014). Dupilumab treatment in adults with moderate-to-severe atopic dermatitis. N Engl J Med, 371(2), 130-139. doi:10.1056/NEJMoa1314768
Berman, H. M., Westbrook, J., Feng, Z., Gilliland, G., Bhat, T. N., Weissig, H., Shindyalov, I. N., & Bourne, P. E. (2000). The Protein Data Bank. Nucleic acids research, 28(1), 235–242. https://doi.org/10.1093/nar/28.1.235
Berman, H., Henrick, K., & Nakamura, H. (2003). Announcing the worldwide Protein Data Bank. Nature structural biology, 10(12), 980. https://doi.org/10.1038/nsb1203-980
Brin M. F. (2023). Entering the 4th decade of Botox® (onabotulinumtoxinA): A narrative review of its development and impact on patients and medicine. Medicine, 102(S1), e32370. https://doi.org/10.1097/MD.0000000000032370
Cockerham, K., & Hsu, V. J. (2009). Collagen-based dermal fillers: past, present, future. Facial plastic surgery : FPS, 25(2), 106–113. https://doi.org/10.1055/s-0029-1220650
Deshmukh SN, Dive AM, Moharil R, Munde P. Enigmatic insight into collagen. J Oral Maxillofac Pathol. 2016 May-Aug;20(2):276-83. doi: 10.4103/0973-029X.185932. PMID: 27601823; PMCID: PMC4989561.
Devore, D., Zhu, J., Brooks, R., McCrate, R. R., Grant, D. A., & Grant, S. A. (2016). Development and characterization of a rapid polymerizing collagen for soft tissue augmentation. Journal of biomedical materials research. Part A, 104(3), 758–767. https://doi.org/10.1002/jbm.a.35599
Ehrlich, M., Rao, J., Pabby, A., & Goldman, M. P. (2006). Improvement in the appearance of wrinkles with topical transforming growth factor beta(1) and l-ascorbic acid. Dermatologic surgery : official publication for American Society for Dermatologic Surgery [et al.], 32(5), 618–625. https://doi.org/10.1111/j.1524-4725.2006.32132.x
Etrusco, A., Geru, M., Laganà, A. S., Chiantera, V., Giannini, A., & Buzzaccarini, G. (2023). Use of botulinum toxin in aesthetic medicine and gynaecology: current approaches, controversies, and future directions. Przeglad menopauzalny = Menopause review, 22(3), 155–160. https://doi.org/10.5114/pm.2023.131457
Fuster Torres, M. A., Berini Aytés, L., & Gay Escoda, C. (2007). Salivary gland application of botulinum toxin for the treatment of sialorrhea. Medicina oral, patologia oral y cirugia bucal, 12(7), E511–E517.
Green, M. (2014, September 25). How Many Units of Botox® is Normal? Dr. Michelle Green, MD Cosmetic Dermatologist. https://www.michelegreenmd.com/how-many-units-of-B...
Intiso, D., Centra, A. M., Gravina, M., Chiaramonte, A., Bartolo, M., & Di Rienzo, F. (2023). Botulinum Toxin-A High-Dosage Effect on Functional Outcome and Spasticity-Related Pain in Subjects with Stroke. Toxins, 15(8), 509. https://doi.org/10.3390/toxins15080509
Kramer, R. Z., Bella, J., Mayville, P., Brodsky, B., & Berman, H. M. (1999). Sequence dependent conformational variations of collagen triple-helical structure. Nature structural biology, 6(5), 454–457. https://doi.org/10.1038/8259
Lacy, D. B., Tepp, W., Cohen, A. C., DasGupta, B. R., & Stevens, R. C. (1998). Crystal structure of botulinum neurotoxin type A and implications for toxicity. Nature structural biology, 5(10), 898–902. https://doi.org/10.1038/2338
Matinong AME, Chisti Y, Pickering KL, Haverkamp RG. Collagen Extraction from Animal Skin. Biology (Basel). 2022 Jun 13;11(6):905. doi: 10.3390/biology11060905. PMID: 35741426; PMCID: PMC9219788.
Noorzai, S., & Verbeek C.J.R. (2020). Collagen: From Waste to Gold. Biotechnological Applications of Biomass. IntechOpen. https://www.intechopen.com/chapters/74308
Scott A. B. (1981). Botulinum toxin injection of eye muscles to correct strabismus. Transactions of the American Ophthalmological Society, 79, 734–770.
Talalaj, U., Uscinowicz, P., Bruzgo, I., Surazynski, A., Zareba, I., & Markowska, A. (2019). The Effects of a Novel Series of KTTKS Analogues on Cytotoxicity and Proteolytic Activity. Molecules, 24(20). doi:10.3390/molecules24203698
Ting, P. T., & Freiman, A. (2004). The story of Clostridium botulinum: from food poisoning to Botox®. Clinical medicine (London, England), 4(3), 258–261. https://doi.org/10.7861/clinmedicine.4-3-258
Trentham, D. E., Townes, A. S., & Kang, A. H. (1977). Autoimmunity to type II collagen an experimental model of arthritis. The Journal of experimental medicine, 146(3), 857–868. https://doi.org/10.1084/jem.146.3.857
Zhao, X. Z., X.; Liu, D. (2021). Collagen peptides and the related synthetic peptides: A review on improving skin health. Journal of Functional Foods, 86. doi:10.1016/j.jff.2021.104680

Covalently conjugating a small molecule to an antibody’s surface is a process called antibody “labeling.” Labeling antibodies with small molecules such as biotin or fluorophores...

DNA gel electrophoresis is one of the most widely used analytical techniques in molecular biology, providing a simple and reliable way to separate DNA fragments...

Do you have a favorite restaurant that you love because you know exactly how great the experience is going to be? There are probably a...

Using the wrong agarose can lead to smearing or poor separation. The good news is that once you know how to evaluate agarose and choose...
