3 Functions of Buffer Solutions in Bioscience Experiments
by Pallabi Roy Chakravarty, Ph.D.

by Pallabi Roy Chakravarty, Ph.D.
Buffers are mainly used three ways. First maintaining pH within a range. Second, buffers supply ions for the flow of electricity. Third, some buffers have chemicals like EDTA, to modify cation concentrations. How buffers work in each of these settings helps us choose and appropriately optimize them.
From the simplest of experiments such as running a DNA gel or maintaining a cell culture, to the most complicated ones, like purifying finicky proteins – regardless of the experiment you are performing, maintaining the pH of the experimental medium is critical.
Since the main function of any buffer is pH maintenance, choosing and using a buffer that is suitable for your experimental needs is a very important part of setting your experiment up.
Keeping this in mind, in the first section of this article, we will talk about how to choose a suitable buffer to maintain the pH desired in your experiment. The theory behind how a buffer restricts the pH within a narrow range is complex. Please take a look at this article – where we have broken down that complicated chemistry so that you can better understand this concept.
In addition to the chemicals that maintain pH, in most cases, buffers also contain some other chemicals such as EDTA. These chemicals are involved with the reduction in the concentration of free cations in the experimental medium, which have specific and sophisticated roles in certain experiments.
Also, ions in the buffer, due to their charge, are responsible for the seamless passage of electric current during electrophoresis.
Using a few specific buffers and their constituents as examples, we will explain how buffers facilitate the flow of electric currents when you want perform protein or nucleic acid electrophoresis.
Buffers maintain pH during the experiment
Buffers determine the concentration of metal ions during the experiment
Buffers provide ions for electric current to flow during electrophoresis experiments
A buffer, in ordinary terms, is something that protects or shields against an unwanted effect. In bioscience, this unwanted effect is a drastic change in pH that might ruin your assay.
The primary function of a buffer solution is to protect against pH fluctuations in the experimental medium.
The scientific mechanism of how a buffer maintains pH is complex, and we have described it in depth in this article.
Maintaining the pH in your experimental solution is essential because all biological components require the right pH for stability and function. Here are some examples to put this into perspective:
This is also the case for blood, which has a very narrow pH range so that the cells and proteins in it can function properly. Even a slight fluctuation in the pH of your blood might land you in the emergency room!
All of these examples show exactly why you would need to carefully maintain the pH of your medium or solution.
The way to easily and carefully maintain the pH is by using a buffer solution. The chemicals in the buffer would maintain the pH of the experimental milieu (or environment) to a very narrow range.
We now come to the most important question: how do you choose the buffer needed for your experiment based on pK value?
You know the pH range needed for your experiment, and the pK of commercial buffers are provided by the manufacturer.
The pK of the buffer you choose should be within +/- 1 of the pH you need to maintain for your experiment.
As a validation of this concept, think of what goes on in biological fluids – most of which need a pH of around 7 to be maintained. The natural buffers in those fluids are ions such as bicarbonate (HCO3-) that has a pK of 6.35, and phosphate (H2PO4-) with a pK of 6.82. If you notice, the pK of these buffering chemicals are 7 +/- 1.
For your experiments, we have a really helpful chart that shows different GoldBio buffers along with their pH ranges and pK values that you might want to check out.
Sometimes, the concentration of metal ions in the experimental milieu is critical. In such experiments, the buffer that is chosen contains chemicals like EDTA that chelate divalent cations and lower their concentration in the solution.

Figure 1. Nucleases require metal ions for
their function. Metal ions bind to nucleases and activate them (left). EDTA
bind to metal ions, and they are no longer available to bind to nucleases and
activate them (right).
An example is Tris-EDTA buffer (commonly TE buffer) for storing nucleic acids.
Let’s imagine that you have isolated DNA or RNA. But before you use them in downstream experiments, you would need to store them properly so that the nucleic acid molecules do not get degraded by nucleases.
Here is where the concentration of cations in the storage solution becomes very important, and we shall see how a buffer like Tris-EDTA (TE) helps with that.
The components of TE buffer are Tris and EDTA.
Tris is involved with the pH maintenance function of this buffer – the concept we just covered in the previous section. For storing DNA and RNA, the pH is maintained at 7.5 and 8, respectively.
Now we get into the EDTA part. EDTA is added to this buffer because it chelates divalent cations such as Mg2+.
Nucleases require these metal ions to function. EDTA binds to these metal ions and because of that – nucleases can no more access them in the storage solution. Without metal ions being available for their functionality, the nucleases become inactive, and your DNA and RNA molecules are safe from degradation.
Also, the pH of 7.5 and 8 are not suitable for DNases and RNases to function, and this further prevents nucleic acid degradation when stored in this buffer.
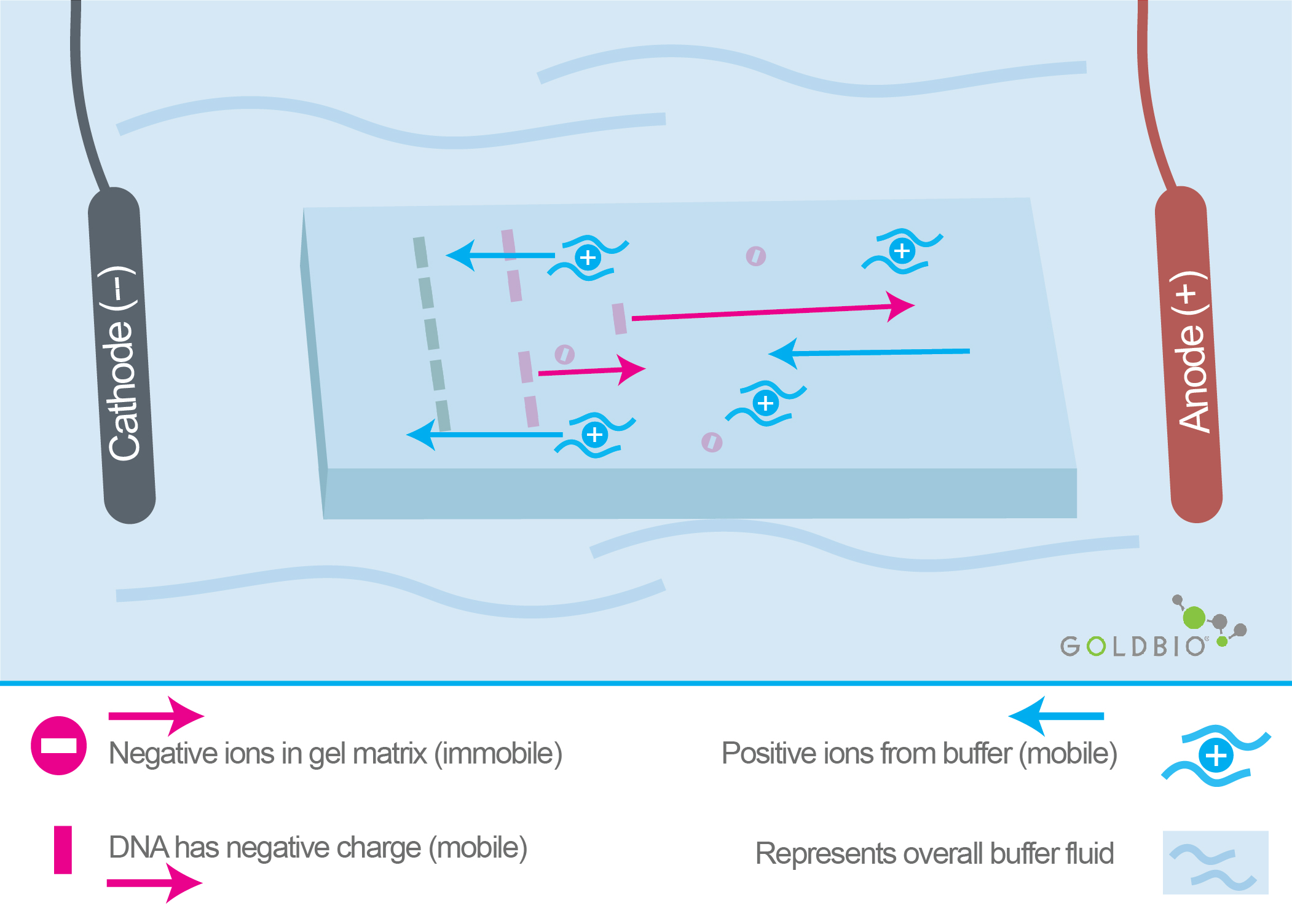
Figure 2.
Representation of a gel for electrophoresis sitting in buffer (TAE). The
positive ions from the buffer are marked with a blue plus sign. Ions in the
buffer help the electric current flow.
During electrophoresis, a constant supply of ions is needed in the gel running solution to ensure the electric current can flow properly.
Two of the most common buffers for running DNA gels are TAE and TBE. Acetic acid (TAE) or boric acid (TBE) is added to TE buffer, respectively.
In addition to maintaining the pH, these weak acids (acetic acid & boric acid) serve the critical function of providing ions by dissociating in the solution. These ions carry the electric current needed for the electrophoresis to take place seamlessly.
Ultimately, we discussed the primary function of buffers and looked at two other important functions that buffers commonly carry out – maintaining the optimal concentration of metal ions and providing ions for electrical currents to flow during electrophoresis experiments.
In addition, specialized buffers have specific components suited for their needs. For example, protein electrophoresis buffers have the detergent SDS to confer an overall negative charge to the polypeptides during electrophoresis, or reducing agents like DTT for breaking disulfide bridges in protein tertiary structure. And bacterial cell lysis buffers may have the enzyme lysozyme that cleaves peptidoglycan in bacterial cell walls.
Nelson, David L. (David Lee), 1942-. (2005). Lehninger principles of biochemistry. New York :W.H. Freeman

Do you ever feel like you’re bouncing through life, squeezing through the never-ending to-do items on your list? If so, then you have a good...

Do you use an AirTag®, Tile®, or other Bluetooth® tracker to keep tabs on anything? When people use these types of tracking devices, it is...

Did you ever get caught passing a note in class? Or the more modern-day equivalent, were you ever caught sending a text message in class?...

Small molecule fluorophores can be attached to biomolecules such as proteins for visualization, tracking, and quantitation in a variety of experiments. There are many small...
