If you find yourself questioning the difference between various luciferase or luciferin types, this guide will set it all straight. Find out what to look out for when shopping for luciferin, why you should choose one type over another, what advantages exist between different luciferins, the importance of purity, storage and more.
GENERAL CONSIDERATIONS
1. Solubility Considerations:
If you just type D-luciferin into our search bar (or any search bar), you’ll get options that include D-luciferin potassium salt, D-luciferin sodium salt and you might see D-luciferin firefly, free acid. So what is the key difference between all three D-luciferin types? Well, for potassium and sodium luciferin there isn’t much difference at all. The two products can be used interchangeably in experiments. However, the sodium salt version is slightly more soluble than the potassium version. And solubility is where the differences really rest, because when it comes to the free acid form of D-luciferin, you’re going to run into some issues. The free acid form is not like the water soluble salts. It requires pH adjustments in order to be made soluble.
Therefore, when it comes to choosing between the three, solubility should be a major consideration.
Note: Why would anyone choose D-luciferin firefly, free acid? Some researchers choose the free acid version despite solubility issues because it contains more photons/milligram than D-luciferin sodium and D-luciferin potassium salts.
|
|
||
2. Assay Considerations:
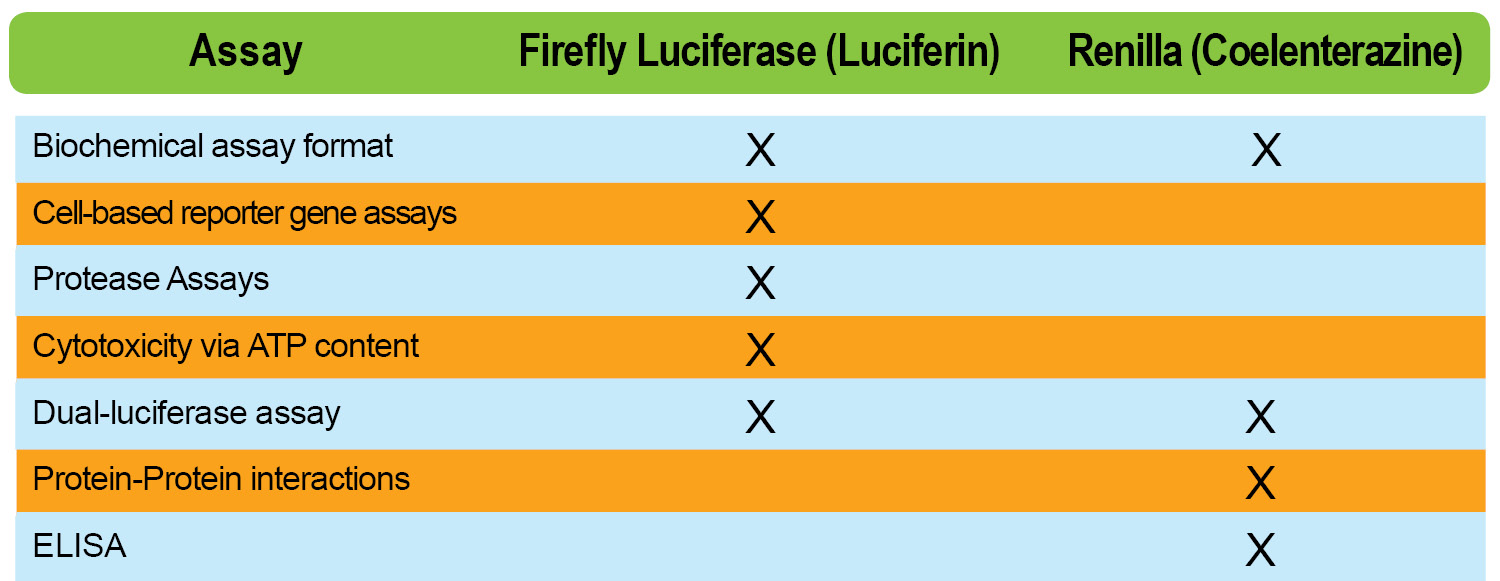
The type of assay you’re working with is going to influence your luciferase choice, which in turn will influence your luciferin choices. So why luciferin over coelenterazine, vice versa or both? Let’s look at it this way:
|
|
||
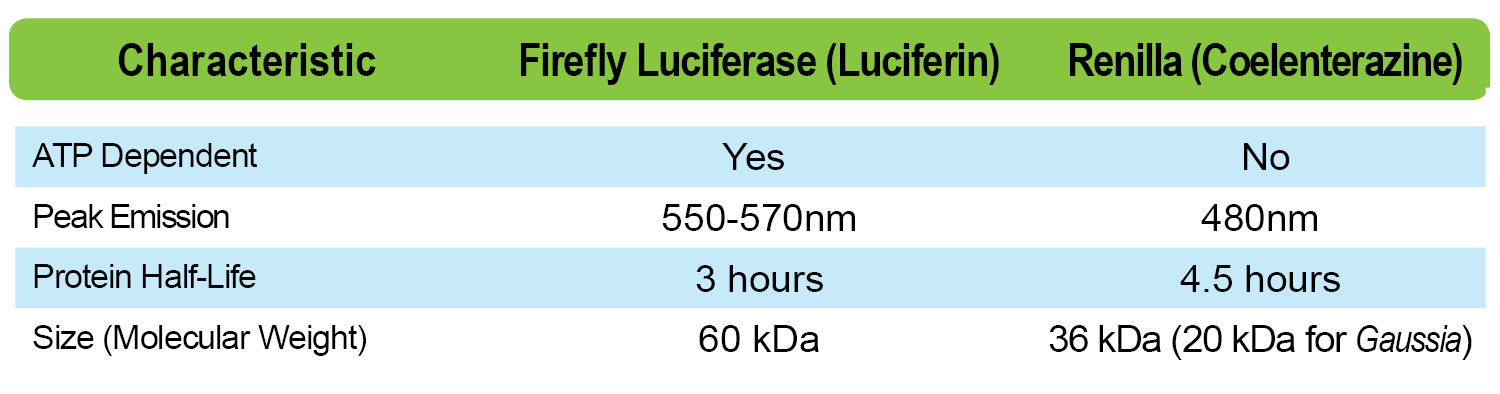
Other things to consider within this topic are some characteristic differences between FLuc (Firefly) and RLuc ( Renilla) such as ATP dependence, peak emission and protein half–life.
|
|
Finally, you need to consider situations that may or may not optimize your experiment. For example, in very small volumes, the FLuc/RLuc dual-luciferase assay is not ideal due to well constraints. Another example, which would lead you to choose coelenterazine instead of D-luciferin has to do with its size. When referring to the chart above, you can see that Gaussia luciferase and Renilla (compatible with coelenterazine) are considerably smaller than available firefly luciferases. In some cases, the advantage of working with a smaller tag is that you can reduce the possibility of interference with your protein of interest, which the tag is attached to.
(Thorne, Inglese & Auld. 2010)
|
|
||
3. Luciferin Types to Consider:
Sodium, Potassium and free acid luciferin were already mentioned previously along with their pros and cons. However, other varieties of luciferin exist such as DMNPE-caged luciferin and L-luciferin. Why would anyone use these – especially L-luciferin? We gathered some important information to answer that question.
a.DMNPE-caged luciferin: Our product description suggests that it can be used to measure intracellular functions (inside the cell) since it readily crosses cell membranes. But what does that mean exactly? For starters, this would suggest that it’s useful for in vivo settings, but then what’s wrong with using D-luciferin potassium or sodium salt? At neutral pHs, D-luciferin runs into some permeability issues. DMNPE-caged luciferin, therefore solves that obstacle by optimizing the approach. Due to its ability to cross the cell membrane, it’s going to allow for the most efficient use of the product.
b.L-Luciferin: L-luciferin is the chiralic sister to D-luciferin and produces no light. While the Lembert paper suggests that it can produce weak levels of light, it is thought that the real reason behind that is the racemization of the chemical back into D-luciferin. So if it’s useless in producing light, what do researchers use L-lucferin for? According to “Bioluminescence: Fundamentals and Applications in Biotechnology, Volume 2,” L-luciferin is helpful in quenching light in dual assay systems. This occurs because L-luciferin acts as a competitive inhibitor, and therefore actually helps increase the Km for D-luciferin, boosting the upper limit for the assay.
|
|
||
|
|
||
4. Coelenterazine Variant Considerations:
We have a fact sheet which highlights information about the three coelenterazine analogues: coelenterazine, coelenterazine h (not available) and coelenterazine 400a. While any of the three are useful in an experiment, this sheet describes reasons why you might want to consider one over the other.
|
|
||
|
|
||
|
|
PURCHASING CONSIDERATIONS
5. Quality & Cost Considerations:
When price shopping for a product, you’re going to encounter a lot of noise. You may find yourself getting a little cross-eyed while perusing offers and sales. Some of them are absolutely worthwhile. However, as you’re weeding through all of these deals, trying to find the best one, be sure you remain vigilant and critical without losing sight of what’s important. Ask yourself about the purity and publication rate. Both are indications about how well you can trust a product. Is a COA available? Can you try a sample?
|
|
||
6. Purity Considerations – How Much Does Purity Actually Matter:
In an earlier article, we explore this topic in greater detail, teaching just how important purity can be, but I’ll pull out one of the more important points in this section.
Deeper into the article we began to talk about what a 99.7% purity really means and compared that to 98%. A 1.7% difference, after doing the math proves to have 0.02 g of potential contaminants. While that doesn’t sound too significant, we find that it equates to 0.8 g per liter, and if these little guys had a molecular weight of 1000g/mol, the concentration would be 800µM!
|
|
||
7. Considerations for What Impurities Might Exist:
Since we're on the topic of purity, it's good to have knowledge of what that means exactly in terms of luciferin. One impurity would be the small presence of L-luciferin, which is a competitive inhibitor. Dehydroluciferin is another inhibitor which can greatly impact the results of your experiment. While you can shop for high-purity luciferins, it is important to properly store and handle the products because it is believes that improper storage can result in the formation of dehydroluciferin.
|
|
||
|
|
||
|
|
STORAGE & HANDLING CONSIDERATIONS
8. Proper Storage of Luciferin:
Essentially, you need to protect your luciferin from light, air and moisture. Oxidation of luciferin is catalyzed by light and oxygen and exposure can rapidly render it useless. Our COAs suggest storing the product desiccated at -20 ˚C in order to maintain the longest shelf life.
|
|
||
9. Proper Handling of Luciferin:
Once you have opened it and are ready to use it, allow the bottle to come fully to room temperature. And before resealing, it must be sparged (removing dissolved gases such as oxygen) with nitrogen or argon.
|
|
||
10. Stock Solution Considerations:
Your prepared stock solutions can be used immediately. And with proper storage, you can keep your stock solution for up to a month. To properly store your luciferin stock, aliquot it out and store it at -80 ˚C.
|
|
||
|
|
||
|
|
BONUS INFO
11. Considerations For Other Luciferases:
In this article, we focused almost entirely on firefly and Renilla luciferases. However, there are other luciferases available for the lab such as Gaussia and click beetle. So we will spend just a little time talking about these luciferases:
a . Click Beetle Luciferase: When talking about what click beetle luciferase is, do not confuse this with “beetle luciferin.” In this case, click beetle luciferase is exactly that, and is derived from Pyrophorus plagiophthalamus. It is particularly useful for dual color assays. Here are the specs for click beetle luciferase:
i. Molecular Weight: 60 kDa (matches firefly luciferase)
ii. Peak Emission Wavelength: Red: 613 nm, green: 537 nm (based on click beetle type)
iii. Substrates: D-luciferin (does not have to be “beetle luciferin”) and ATP
iv. Protein Half-Life: 7 Hours
v. Uses: Primarily for dual color assays, allowing for simultaneous monitoring of a variety of events due to their different color emissions.
b. Copepod Crustacean (Gaussia): This luciferase type was mentioned earlier. It is derived from marine copepod and produces stronger flash signals than firefly luciferase and Renilla luciferase. Here are the specs for Gaussia luciferase:
i. Molecular Weight: 20 kDa
ii. Peak Emission Wavelength: 460 nm
iii. Substrates: Coelenterazine (no ATP dependence)
iv. Protein Half-Life: 6 days in cell media
v. Uses: It can be used for live cell assays, and its small size offers an advantage by reducing the interference with the protein of interest.
c. Ostracod Crustacean (Vargula/ Cypridina): This is another marine version and is also known as the sea firefly. It isn’t nearly as common as firefly luciferase and therefore information about this particular type is far more limited:
i. Molecular Weight: 62 kDa (matches firefly luciferase)
ii. Peak Emission Wavelength: 465 nm
iii. Substrates: Vargulin also known as Cypridina luciferin (no ATP dependence)
iv. Protein Half-Life: 53 hours in cell media
v. Uses: Its sensitivity and detection of small amounts of luciferase activity is one advantage as well as its ability to be multiplexed with other luciferases. It is used for promoter studies, drug screenings and signal transduction pathway analysis.
(Thorne, Inglese & Auld. 2010)
|
|
||
References:
Lembert, Nicolas. "Firefly luciferase can use L-luciferin to produce light." Biochemical Journal 317.Pt 1 (1996): 273.
Luque-Ortega, J., Rivero-Lezcano, O., Croft, S., Rivas, L. (2001). In vivo monitoring of intracellular ATP levels in Leishmania donovani promastigotes as a rapid method to screen drugs targeting bioenergetic metabolism. American Society for Mircrobiology. Doi: 10.1128/AAC
Nakamura, Mitsuhiro, et al. "Construction of a new firefly bioluminescence system using L-luciferin as substrate." Tetrahedron letters 47.7 (2006): 1197-1200.
Ohmiya, Y. (2014). Applications of bioluminescence: Cell based assays and imaging. Photobiological. Retrieved March 11, 2016 from http://www.photobiology.info/ohmiya.html
Thousand, G., & Marks, R. (n.d.). Bioluminescence: fundamentals and applications in biotechnology (Vol.2). New York City, NY: Springer.
Thorne, N., Inglese, J., & Auld, D.S. (2010). Illuminating insights into firefly luciferases and other bioluminescent reporters used in chemical biology. Chemistry & Biology, 17(6), 646-657. Retrieved March 15, 2016, from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2925662/
 Karen Martin GoldBio Marketing Coordinator |
"To understand the universe is to understand math." My 8th grade math teacher's quote meant nothing to me at the time. Then came college, and the revelation that the adults in my past were right all along. But since math feels less tangible, I fell for biology and have found pure happiness behind my desk at GoldBio, learning, writing and loving everything science. |
Category Code: 79104 88251






