Western Blot Deep Dive: What It Is and How It Works
by Pallabi Roy Chakravarty, Ph.D.

by Pallabi Roy Chakravarty, Ph.D.
Western blot, also called immunoblot, is a commonly used technique for qualitatively and quantitatively analyzing proteins in an experimental sample.
Basically, Western blot tells you whether your protein of interest is present in the sample and by how much in relative abundance.
Western blot involves resolving out the proteins in the experimental sample by gel electrophoresis, transferring the electrophoresed proteins from the gel onto a membrane, and then detecting the target protein of interest immobilized on the membrane using antibodies that act as molecular probes. Finally, the membrane is developed using detection reagents for visual comprehension of the results.
The transfer of the proteins from the gel to the membrane gives the name “blot,” just as spilled juice is blotted off from your dining table using a paper towel.
Basic steps of a Western blot experiment are:
Indeed, protein analysis is a cornerstone in modern bioscience.
Western blot plays a big role in protein biochemistry research.
In this article, we will look at the theoretical concepts underlying each step in a Western blot. Some variations in Western blot will also be discussed.
Article Contents:
Why is it called Western blot?
Sample preparation for Western blot
Gel-to-membrane transfer of proteins
Basic principle of Western blot transfer
A note about immunoblot (Western blot) membranes
Steps in the Western blot Transfer
The actual detection process in a Western blot
Probing the membrane with primary and secondary antibodies
Final detection assay: developing the immunoblot for visual analysis
Western blot got its name from another, earlier discovered blotting technique called Southern blot (for DNA analysis) named for its discoverer Edwin Southern.
Since then, other developed blotting techniques have been named for the cardinal directions, just for keeping synchrony in the nomenclature of blotting techniques: Northern for RNA and Western blotting for protein analysis respectively.
Samples for Western blot analysis may come from plant or animal tissue, for example, to study whether a specific protein is being expressed in a transgenic spinach leaf or in the mouse ear.
Or, the samples might also come from a cell or microbial culture.
Samples may even be clinical. For example, a clinical scientist might want to know whether a specific protein is present in the patient’s blood or in an excised tumor.
Figure 1 depicts this step.

Figure 1. Depiction of where protein samples my originate from for extraction and analysis.
For some insight into common approaches for protein extraction and isolation from the source its sample (cell line, tissue, microbial culture etc.) please refer to this article.
For Western blot analysis, proteins in the sample, in most experiments, are denatured to their primary structure by the action of heat, the detergent Sodium Dodecyl Sulfate (SDS), and reducing agents such as beta mercaptoethanol (BME) or dithiothreitol (DTT).
However, it is critically important that the entire polypeptide chain for all proteins in all of the samples that will be Western blotted are preserved and protected from breaking down.
If the polypeptide chain is broken down or truncated, Western blot results will be erroneous.
For this, during protein extraction and isolation steps, great care needs to be taken that the polypeptides are not broken down by the action of proteases.
Proteases are enzymes that breakdown proteins.
Proteases might be in your sample from the beginning. For example, your protein sample is from a living tissue and proteases are there in most cells.
Proteases might also get into your sample by accidental contamination. For example, if you are talking to a labmate during protein extraction and a speck of your saliva or air droplets, which contains proteases, might get into your sample and then degrade the polypeptides.
Proteases, if contaminating the experimental protein sample, may cause lysis or breakdown of the proteins (proteolysis). This, of course, is an unwanted situation. There is a technical term for this – ‘protease-mediated unwanted proteolysis.’ In other words, it's the lysis of your proteins caused by proteases, and we didn't want that to happen.
To avoid unwanted protease-mediated proteolysis, inhibitors of specific protease types or cocktails of multiple protease-inhibitors are used during protein extraction and sample preparation for Western blot. Please refer here for a wide consortium of protease inhibitors for your experiment.
After sample preparation, the second step in a Western blot experiment is electrophoresing the protein samples in a gel.
The whole purpose of electrophoresis is to separate out the target protein of interest from the rest of the proteins in each sample so you can detect it properly.

Figure 2. Electrophoresing protein samples.
In figure 2, four lanes are being loaded. One is the protein ladder. Consider the samples in tubes 1, 2 and 3 as proteins extracted from three different genetic mutants of a bacterial strain. Tube 4 is a control, that is, proteins extracted from the laboratory strain of the bacterium in which these 3 mutations (samples 1-3) were made. You want to study how a specific toxin protein is expressed because of these mutations, as compared to that in the non-mutant strain.
Since the basis of this separation in most Western blot experiments is molecular weight, the most common electrophoresis method followed for Western blot is SDS-PAGE.
SDS-PAGE involves denaturing proteins in the sample to their primary structure – all higher levels of protein structure that mediate protein-protein interactions are destroyed.
For preserving protein-protein interactions and analyzing protein complexes as opposed to single polypeptides in a Western blot, non-denaturing electrophoresis methods such as native PAGE are used instead of SDS-PAGE.
In most immunoblots, protein ladders are loaded in a well during electrophoresis.
As shown in Figure 3 below, following electrophoresis the gel typically looks like this. The ladder bands are visible but none of the actual proteins in the sample are yet visible.

In this context, here is a quick note on the main advantages of running a colored protein ladder in a Western blot:
* Since the ladder is visible in the gel, it helps you visually confirm how far your proteins likely ran through the gel. Also, the ladder shows an estimate of how much separation has taken place between proteins of different molecular weights.
* After the transfer step that follows electrophoresis, the colored bands of the ladder will help you guess the efficiency of protein transfer from the gel to the membrane.
Following electrophoresis, the next step in an immunoblot is transferring the proteins from the gel to a membrane. After transfer to the membrane, the electrophoresed proteins are immobilized for detection and further analysis.
Here is an analogy to help understand the blotting step.
Think about a paper towel used to soak up or absorb, or ‘blot up’ spilt liquid (for example, orange juice) from your dining table.
So also, the proteins are blotted onto the Western blot membrane from the electrophoresed gel. The gel is analogous to your dining table, the spilt water akin to the protein samples and the paper towel is the membrane.
If the protein sample is blotted directly onto the membrane without electrophoresis, it is a variation of Western blot called dot blot.
Like a dot – opposed to separated bands from an electrophoresed gel, the sample is blotted onto the membrane.
Dot blot is a quick version of Western blot just to see if your target protein is indeed present in the sample. If yes, then generally a full course Western blot is performed on the sample for detailed analysis.
All protein samples have an overall negative charge due to pre-treatment with SDS before electrophoresis. Because of this, proteins migrate towards the positive electrode whenever exposed to an electric current.
Utilizing this, the basic transfer setup is shown in Figure 4.
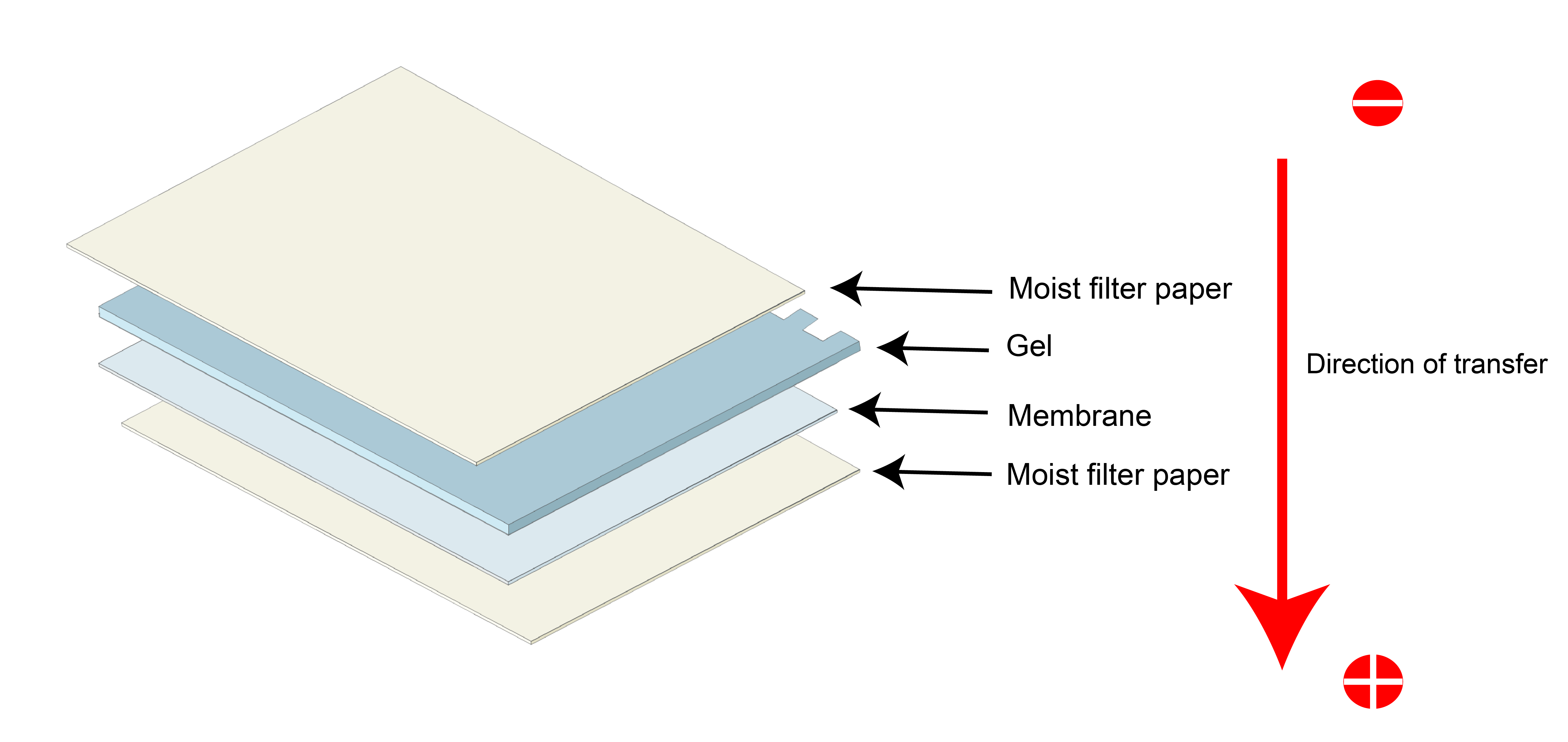
Figure 4. Basic principle of the transfer. Negatively charged proteins from the gel (which is towards the (-) electrode flow to the membrane (which is towards the (+) electrode).
The gel is placed directly on the membrane.
Importantly, the gel is placed on the side of the negative and the membrane adjacent to the positive electrode respectively.
Filter papers are placed between the positive electrode and the membrane, and between the gel and the negative electrode.
Since the entire transfer is dependent on the flow of electric current, the whole transfer setup is moistened in a transfer buffer.
A transfer buffer ensures that an electric current can flow easily, and in the process, the proteins get transferred from the gel to the membrane.
Now that we understand the basic principle of transfer, here are the steps of a Western blot transfer:
Step 1: The gel is processed following electrophoresis. This is illustrated in Figure 5.

Figure 5. Processing of the gel for blotting
Gel processing following electrophoresis involves cutting off and discarding the stacking portion from the top of the gel. The stacking gel does not contain any protein sample or ladder following electrophoresis.
The resolving gel is carefully put into a tray of transfer buffer. Three things need to be ensured at this point:

The membrane comes enwrapped within two sheets of covering (shown in light blue color). It is cut according to the size of the gel, taken out from within the 2-layer paper wrap with a pair of forceps and put in a tray of transfer buffer for equilibration.
The filter papers are also cut out similarly and equilibrated in transfer buffer.
Like the gel, the membrane is also not touched by bare hands to avoid any protein contamination.
Step 3: The transfer sandwich is made, as shown in Figure 6.
Following transfer, a membrane typically looks like what is illustrated in Figure 7.

Figure 7. Represents how a membrane looks after transfer.
The ladder lane shows up, but the actual samples transferred onto the membrane cannot yet be visualized.
There are two methods of transfer – wet and semi-dry.
In wet transfer, the transfer sandwich is placed in a tank full of transfer buffer. This transfer tank has two electrodes through which current flows.
The sandwich is placed between two sponges as shown in figure 8. The orientation is: sponge, filter papers, gel, membrane, filter papers, sponge – fitted within a cassette in this order. This cassette is inserted in the tank of buffer.

Figure 8. Western blot wet transfer sandwich cassette.
In the semi-dry method, the transfer sandwich – filter papers >> gel >> membrane >> filter papers, moistened in transfer buffer is placed directly between the electrodes.
The sandwich is moist, but unlike the tank (wet transfer) method, it is not immersed in transfer buffer.
Think about the architecture of a semi-dry transfer analogous to the process of making a pressed sandwich.
The two surfaces of the sandwich-maker are your positive and negative electrodes. The transfer sandwich is akin to your sandwich – the filter papers on the two sides are the slices of bread, the gel is your slice of deli meat and the membrane is the slice of cheese. The whole sandwich is moistened in gravy, I mean the transfer buffer.
Wet transfer is more efficient in transferring proteins, especially larger ones.
The tank has a lot of buffer, which ensures smooth and constant flow of the electrical current during the whole transfer.
Also, the tank can be kept cool by doing the transfer in a cold room. This mitigates the problems that arise due to the buffer and the sandwich heating up during transfer, due to the flow of electric current.
In the semi-dry, there are chances of the sandwich drying up or over-heating during transfer.
Because of these factors, consider the wet method as sophisticated, slow-cooking to perfection.
On the other hand, the semi-dry method is cheaper because it uses far less transfer buffer, is faster and is less cumbersome.
After transfer, comes protein detection. This involves three main steps: blocking the membrane, treatment with antibodies and developing the membrane for final detection and visualization.
Often, the step of staining the membrane for total protein estimation is done prior to blocking.
Using a colored protein ladder lets you somewhat track efficiency of transfer – the more the intensity of the colored bands, the better the transfer.
But this may just be for the ladder lane. You do not know how well samples in other lanes have transferred. If transfer efficiency between lanes (inter-lane) and within a lane (intra-lane) differ, it may lead to erroneous results.
A way around this problem is to stain the whole membrane.
There are multiple stains sold by GoldBio for membrane staining. A representative image of what the membrane looks like after staining is shown in Figure 9.
 Figure 9. Rendered example of what a stained Western blot membrane could look like.
Figure 9. Rendered example of what a stained Western blot membrane could look like.
Whole membrane staining is used mainly for two purposes:
1)to track transfer efficiency.
2)to have loading controls.
Though a little beyond the scope of this article, here is a small note on loading control in a Western blot.
Loading control means you show evidence that an equal amount of total protein is loaded in each lane. This is important in proving that if your target protein shows up with differential abundance in different lanes (having different samples), it is due to a biological effect and not due to differential loading.
After the optional step of total protein staining, you initiate the actual detection steps starting with blocking the membrane.
Prior to the antibody treatments for detecting the target protein, the membrane is ‘blocked’ with a protein solution such as bovine serum albumin (BSA) or non-fat dry milk.
This prevents non-specific binding, or noise.
The method and utility of blocking is explained in Figure 10.

Figure 10. The utility of blocking a Western blot membrane. The primary antibody can bind to the target protein (in dark blue) due to its specific and very high affinity. The rest of the non-target proteins cannot be bound by the primary antibody because the blocking solution blocks all non-specific binding of the primary antibody. This improves signal:noise ratio of the western blot results.
The primary antibody serves as your probe in detecting your target protein on the membrane.
On the other hand, the secondary antibody serves as the probe to detect the primary antibody – bound to the target protein on the membrane.
For this reason, treating the membrane with antibodies for detection is called probing.

Figure 11. Target protein detection in a Western blot using a primary and a secondary antibody.
After secondary antibody treatment, the final step is the detection assay and developing the membrane.
Depending on the type of detection method that your secondary (or primary antibody, if no secondary antibody is involved) is conjugated to, the detection assay may be any one of these methods:
One of these 3 methods is conjugated to the antibody probe, as shown in Figure 11.

The protein streptavidin clamps onto the small molecule biotin in one of the tightest natural interactions ever discovered. Over the years, the strength of this...

Maybe you are about to start working with D-luciferin, and you begin reviewing protocols. But what you’re doing is a little different. Or what you...

Handles are common in everyday life. We use them to help us open doors, cabinets, or to hold coffee mugs, cookware, and tools. They are...

Labeling antibodies with biotin or a fluorophore enables scientists to detect, track, and quantify that antibody and the molecules that it binds to. We cover...
