Ethidium bromide (EtBr) has been the go-to product for nucleic acid staining; however, you’re probably well aware of the risks involved in using this chemical. One of the more popular, safe alternatives is GelRedTM, which GoldBio carries. If you have been doing any kind of research on this product or other alternatives, this article will help answer some of your big questions about GelRedTM sensitivity, safety, usage and troubleshooting.
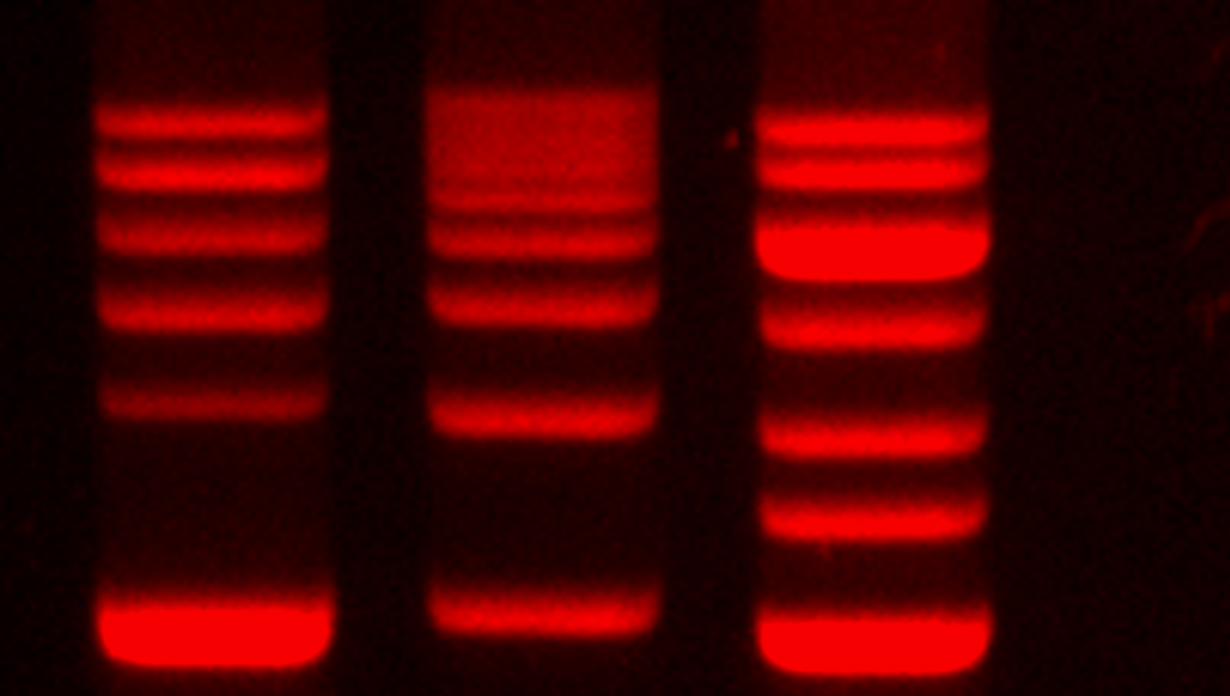
GelRedTM is a highly sensitive fluorescent nucleic acid dye that works when excited by a transilluminator. It was designed to replace EtBr, which is considered to be carcinogenic and mutagenic. GelRedTM is also known to be very sensitive without requiring a destaining step. In order for manufacturers to be sure its design is safe, GelRedTM was subjected to various tests, which concluded it is nontoxic, nonmutagenic and nonhazardous, even at very high concentrations. GelRedTM has the advantage of better safety with less disposal headaches while still providing results that are functionally similar to EtBr.
With all these advantages, what are the disadvantages? Is it too good to be true? To answer that, let’s look at some of the most common questions people have before or while they’re working with GelRed
TM:
|
|
||
How does GelRed Work?
Like ethidium bromide, GelRedTM is a nucleic acid dye that intercalates with DNA. And it binds exclusively by intercalation.
When UV light is shown, the intercalated GelRedTM will fluoresce producing a bright orange light. Both products have nearly the same spectra; however, GelRedTM is considered a much safer alternative to EtBr and is much more sensitive. Another benefit of GelRedTM is that it is able to work with dsDNA, ssDNA and RNA, and when it comes to dsDNA, it is twice as sensitive as ssDNA and RNA.
|
|
||
If GelRed is an intercalating agent why doesn’t it have the same risk as ethidium bromide?
This is really a great question! It seems logical to wonder how it can truly be safe if it’s still going to bind directly with DNA. While GelRedTM and EtBr both intercalate with DNA, GelRedTM was created so that it does not penetrate the cell membrane. Because it cannot penetrate the cell membrane, GelRedTM is not toxic to living cells.
When GelRedTM and GelGreenTM were developed, scientists focused on creating a product with three major features in mind: it would not be able to penetrate latex gloves, it would not be able to penetrate the cell membrane, and it would be metabolized into compounds that have little interaction with DNA.
To ensure GelRedTM and GelGreenTM accomplished these goals, GelRedTM and GelGreenTM were tested using the glove penetration test, the Ames test, the cell permeability and cytotoxicity test and the environmental safety test.
|
|
||
How does GelRed compare to ethidium bromide?
To answer this question, let’s compare GelRedTM to EtBr in a few different ways:
- Safety
- Procedure
- Cost
- Sensitivity
- DNA Migration
Already, the chart below can start to help visualize aspects of the comparison. GelRedTM is clearly known to be safer to use than EtBr, but it also has an equal (if not better) level of sensitivity, excels in disposal ease, but does come up short when it comes to cost.
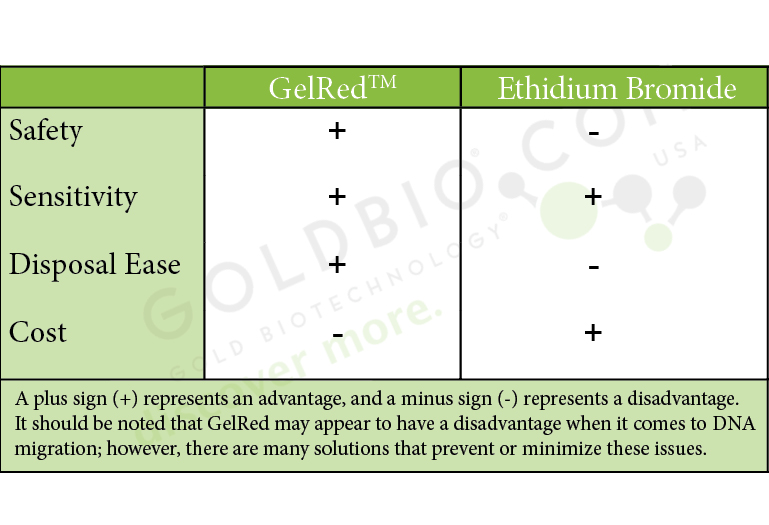
Let’s get a deeper look at how these two products compare…
Safety of GelRedTM vs. Ethidium Bromide:
Right away, the most obvious advantage GelRedTM has over ethidium bromide is the safety, especially when it comes to GelRedTM Nucleic Acid Gel stain, 10,000X in water. Because of its safety, GelRedTM is easily disposed directly into the drain. One warning when it comes to disposal, however, is to contact your safety office about local disposal guidelines.
Procedure using GelRedTM vs. Ethidium Bromide:
Another great advantage of GelRedTM is that there is no need for a destaining step. Unlike EtBr, which can sometimes generate background without destaining, GelRedTM provides very clear images
Cost of GelRedTM vs. Ethidium Bromide:
Cost is immediately seen as a disadvantage of GelRedTM over EtBr. However, researchers should factor in the cost and time required for proper disposal of EtBr. When considering the indirect factors along with safety, GelRedTM still offers considerable benefits.
Sensitivity of GelRedTM vs. Ethidium Bromide:
Both GelRedTM and ethidium bromide are very sensitive stains, especially compared to other alternatives on the market.
DNA migration using GelRedTM vs. Ethidium Bromide
The affect GelRedTM has on DNA migration is a common concern among researchers. While EtBr has little to no impact on migration, in certain circumstances, GelRedTM can hinder it. Though migration is a concern, it is not necessarily a disadvantage since there are ways to significantly decrease the impact GelRedTM has on migration (see further in this article for more details).
|
|
||
What are the best conditions to use for GelRed staining? How do you prevent blurred DNA bands? And what filter should I use for GelRed?
Conditions/optimization for GelRedTM
The first big way to make sure you’re optimizing your procedure is to do a post-stain with GelRedTM rather than a precast gel. A post-stain will reduce the risk of GelRedTM interfering with DNA migration and provide you with the best sensitivity. If you are working with polyacrylamide gels, then avoid a precast stain altogether since it is not recommended.
If you have performed a precast stain and are getting blurred bands, you can try a few things to troubleshoot this issue:
- Reduce the amount of DNA loaded by one-half to one-third (<400ng). GelRedTM is more sensitive than ethidium bromide. Smeared bands are usually the result of overloading, which is commonly observed with DNA ladders.
- Switch to a post-stain rather than a precast stain.
- Try pouring a lower percentage gel, especially when working with larger fragments.
- If you are using TAE buffer, try TBE instead. TBE has a higher buffering capacity than TAE.
See the GelRedTM protocol for more troubleshooting tips.
Emissions Filter for GelRedTM:
An advantage with GelRedTM is its similarity to EtBr. Therefore, you can use the ethidium bromide filter when you’re working with GelRedTM. You can also use the SYBR® filter or the GelStarTM filters during imaging and get great results.
|
|
||
Why does GelRed alter the migration of DNA?
There are a number of reasons why GelRedTM affects the migration of DNA. Remember, that all nucleic acid dyes will have a certain impact on the way DNA moves. GelRedTM is a much heavier dye than EtBr, and GelRed’s affinity for DNA means that the more DNA that is loaded in a gel, the more GelRedTM will bind to DNA and skew the movement of that fragment.
A few recommendations to prevent or minimize the impact GelRedTM has on migration are: use it as a post-stain (we cannot stress this enough). Be sure your GelRedTM stock solution is appropriately diluted. You may also need to reduce the amount of DNA you’re using to minimize the impact of migration interference.
|
|
||
What’s the difference between GelRed and GelGreen?
The first primary difference is the fluorescence from each product. GelRedTM will, as you would expect, give off a red fluorescence much like ethidium bromide. And GelGreenTM, as you would expect, gives off a green fluorescence much like SYBR® Green. Either product can be used with a UV transilluminator.
Another difference is staining sensitivity. GelRedTM is a more sensitive stain (about five times more sensitive) than GelGreenTM, especially for single-stranded nucleic acids.
In terms of application, GelRedTM is compatible for with polyacrylamide gels, whereas that is not recommended for GelGreenTM.
Finally, one advantage of using GelGreenTM is its compatibility with blue light transilluminators, which is helpful if you want to avoid ultra violet exposure.
|
|
||
What’s the difference between DMSO GelRed and water based GelRed?
We offer both GelRedTM and GelGreenTM products in DMSO or water. It is recommended, however, to choose the water formulation which helps to prevent any potential hazards in use since DMSO is able to be absorbed into the skin. Both options are still available to serve researchers who have longstanding protocols that use the DMSO formulation.
|
|
||
Is GelRed compatible with ssDNA and RNA? Is it compatible with Southern blot?
GelRedTM is definitely compatible with single-stranded DNA as well as RNA. However, its sensitivity is two times greater for double-stranded DNA.
When it comes to the Southern blot application, GelRedTM is compatible. But it is highly recommended to use the post-stain protocol if you are doing a blotting application.
|
|
||
What downstream applications is GelRed compatible with?
GelRedTM is structurally similar to ethidium bromide and is compatible with downstream DNA applications including restriction digest, ligation, sequencing and cloning. GelRedTM is also compatible with cesium chloride gradients, use in Southern blot applications and the Comet assay (single cell gel electrophoresis).
Another benefit of GelRedTM is its compatibility with the UV transilluminator, which requires no change in setting; you can use the ethidium bromide filter for GelRedTM. The SYBR® and GelStarTM filters are also compatible with GelRedTM.
|
|
||
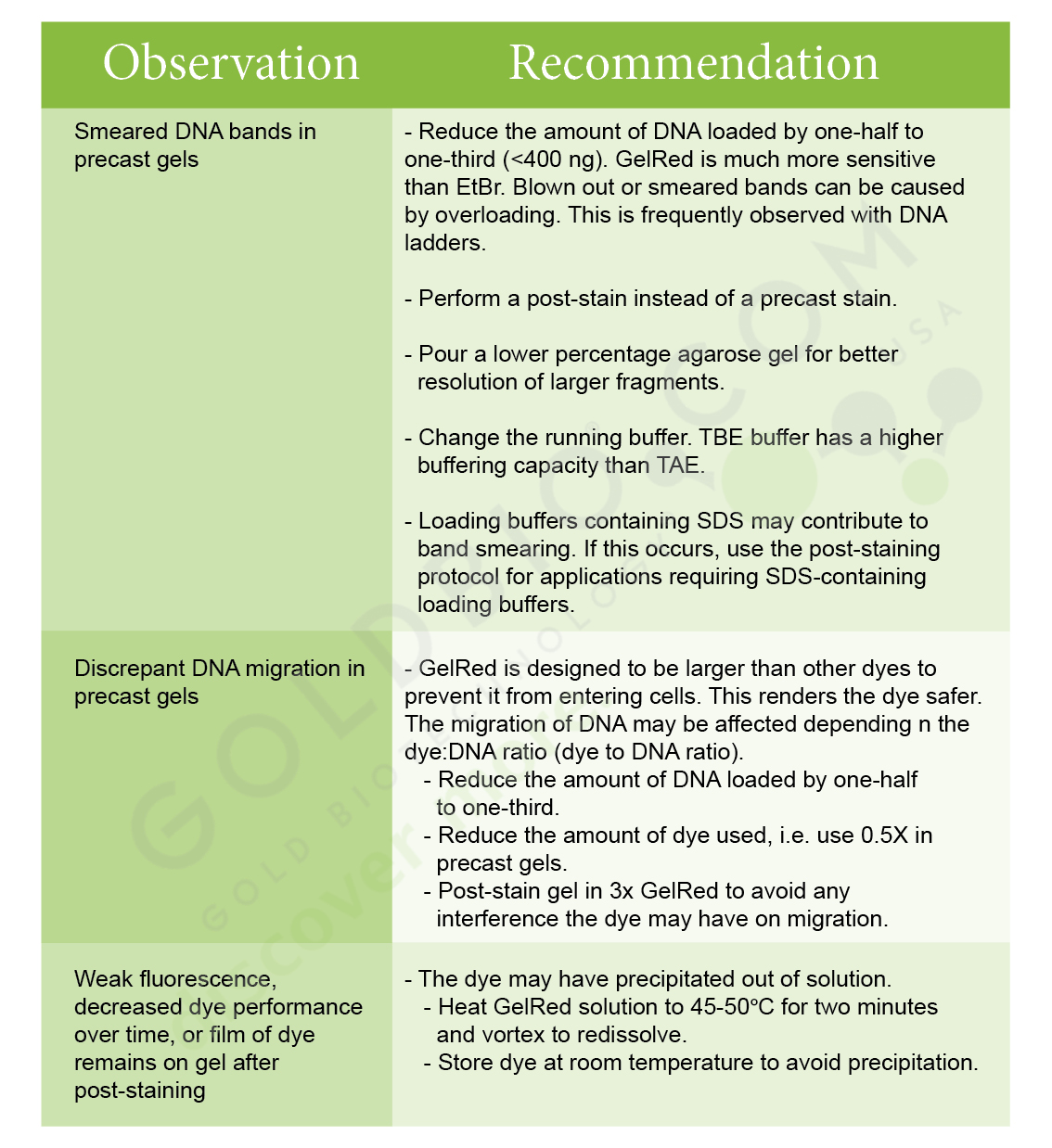
How do I troubleshoot issues with GelRed?
Our GelRedTM Nucleic Acid Gel Stain, 10,000X protocol has a lot of troubleshooting information, which is particularly helpful if you’re experiencing issues such as smeared bands in a precast gel, discrepant DNA migration in precast gel or weak fluorescence. Below is the troubleshooting table from the protocol.
|
|
||
References:
Ames, Bruce N., and Lois S. Gold. "The Causes and Prevention of Cancer: The Role of Environment." Biotherapy11 (1998): 205-220.
"Ames Test." Genetics. . Retrieved February 27, 2017 from Encyclopedia.com:http://www.encyclopedia.com/medicine/medical-magazines/ames-test
Berge, T., Jenkins, N. S., Hopkirk, R. B., Waring, M. J., Edwardson, J. M., & Henderson, R. M. (2002). Structural perturbations in DNA caused by bis-intercalation of ditercalinium visualised by atomic force microscopy. Nucleic Acids Research,30(13), 2980-2986. Retrieved February 15, 2017, from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC117064/.
Crisafuli, F. A., Ramos, E. B., & Rocha, M. S. (2015). Characterizing the interaction between DNA and GelRed fluorescent stain. European Biophysics Journal,44(1), 1-7. doi:10.1007/s00249-014-0995-4
EDVOTEK. (n.d.). EDVOTEK Quick Guide: Visualizing DNA[Brochure]. Washington DC. Retrieved February 14, 2017.
Huang, Q., Baum, L., & Fu, W. L. (2010). Simple and practical staining of DNA with GelRed in agarose gel electrophoresis. Clinical Laboratory,56, 149-152. Retrieved February 14, 2017.
Kimball, J. (2010, December 21). Ames Test [Web blog post]. Retrieved February 14, 2017, from http://www.biology-pages.info/A/AmesTest.html
Kirkwood Community College Math/Science Department (Producer). (2012). Gel Red vs. EtBr [Video file]. Retrieved February 10, 2017, from http://www.benchfly.com/video/158/gel-red-vs-etbr/
Menne, C. (2017, January 13). GelRed Nucleic Acid Gel Stain FAQ [PDF] St. Louis: Gold Biotechnology.
Menne, C. (2017, January 13). GelRed Nucleic Acid Gel Stain, 10,000X Procedure for staining dsDNA, ssDNA or RNA in gels [PDF] St. Louis: Gold Biotechnology.
Oswald, N. (2008, March 03). Ethidium Bromide: The Alternatives [Web blog post]. Retrieved February 15, 2017, from http://bitesizebio.com/417/ethidium-bromide-the-al...
Product Information [PDF]. (2013, June 13). Hayward: Biotium.
“PulseNet: Under the Microscope” Volume 2 Alternate DNA Stains – Results and Recommendations [PDF]. (n.d.). PulseNet International.
Safety Report of GelRed and GelGreen[PDF]. (2013, October 16). Hayward: Biotium
 Karen Martin GoldBio Marketing Coordinator |
"To understand the universe is to understand math." My 8th grade math teacher's quote meant nothing to me at the time. Then came college, and the revelation that the adults in my past were right all along. But since math feels less tangible, I fell for biology and have found pure happiness behind my desk at GoldBio, learning, writing and loving everything science. |
Category Code: 88251 79105 79104 79103 79107 79109





