All About Differential Media in Gene Cloning - What it is and How it Works
by Pallabi Roy Chakravarty, Ph.D.

by Pallabi Roy Chakravarty, Ph.D.
Differential media is culture media containing compounds that enable visual distinction between the microbes growing on it. These media leverage the distinct metabolic and biochemical capabilities of microbes, resulting in visible variations in appearance.
Differential media often incorporate indicators such as dyes, pH indicators, or specific substrates that can be metabolized by certain microorganisms, but not by others. The presence or absence of these indicators or the resulting color changes help discern different microbial groups or identify specific metabolic activities.
For example, blue-white screening using differential media containing X-gal facilitates gene cloning by identifying colonies with the desired genetic construct.
How is differential media different from selective media?
Media for blue-white screening
The difference between selective and differential media has to do with how each is used to screen transformed cells. Selective media screens cells based on growth while differential media screens based on appearance.
Both selective and differential media can help you decide which colonies to move ahead with in your cloning procedure.
With selective media, the undesired cells fail to survive. The selection is therefore based on growth.
The typical compound that mediates the differentiation between the desired and the undesired cells is an antibiotic. We have a great article that goes into more detail about selective media and how it works here.
The desired cells have resistance to this antibiotic and can grow even in the presence of that antibiotic. The undesired cells, however, are sensitive to the antibiotic and do not survive.
With differential media, both desired and undesired cells grow, but they can be distinguished and screened based on visual characteristics of the colonies.
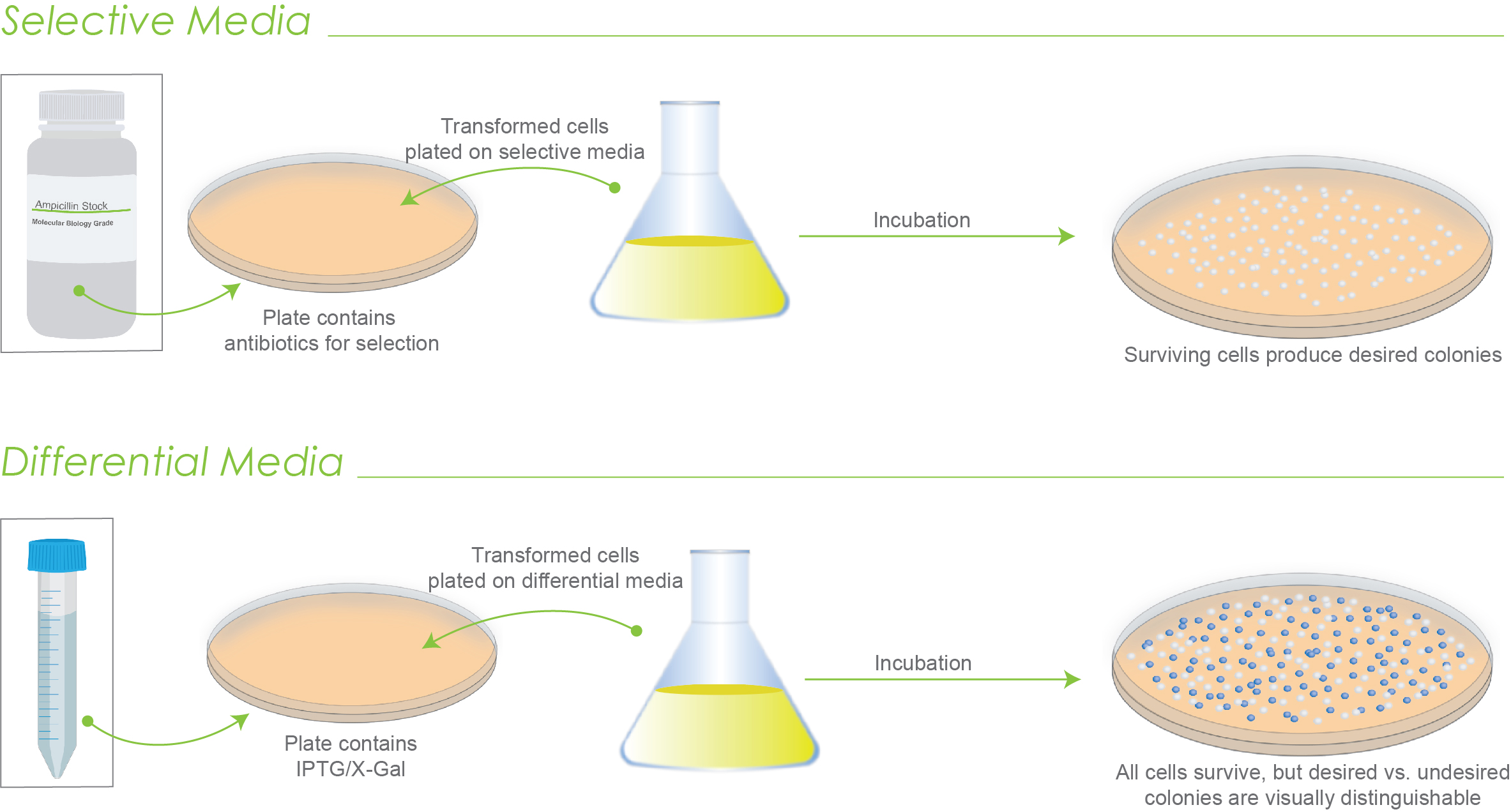
Figure 1. Illustration of how selective media works versus how differential media works. Only surviving colonies grow on selective media. With differential media, all colonies grow, but can be discerned.
The way differential media works is that they contain a substrate for a specific enzyme.
The substrate, once broken down by this enzyme, produces a colored product. The color difference can easily be detected by the naked eye.
Figure one’s example of differential media shows this color difference in the results where white colonies are desired colonies, and blue colonies are not.
To put this more into perspective, look at figure 2 which demonstrates how this works.
You add a certain compound (white in color), we’ll call compound C in your solid media plates. When compound C is broken down by a specific enzyme, which we will call enzyme E, the product is a blue compound.

Figure 2. Conceptual representation of how differential media works.
Now, in this specific example, you design your genetic construct in such a way that the cells that have the desired gene construct will produce enzyme E. And so, their colonies will be blue once they grow on a plate.
On the other hand, the undesired colonies – the ones without the desired genetic construct will not produce enzyme E, and their colonies will be white.
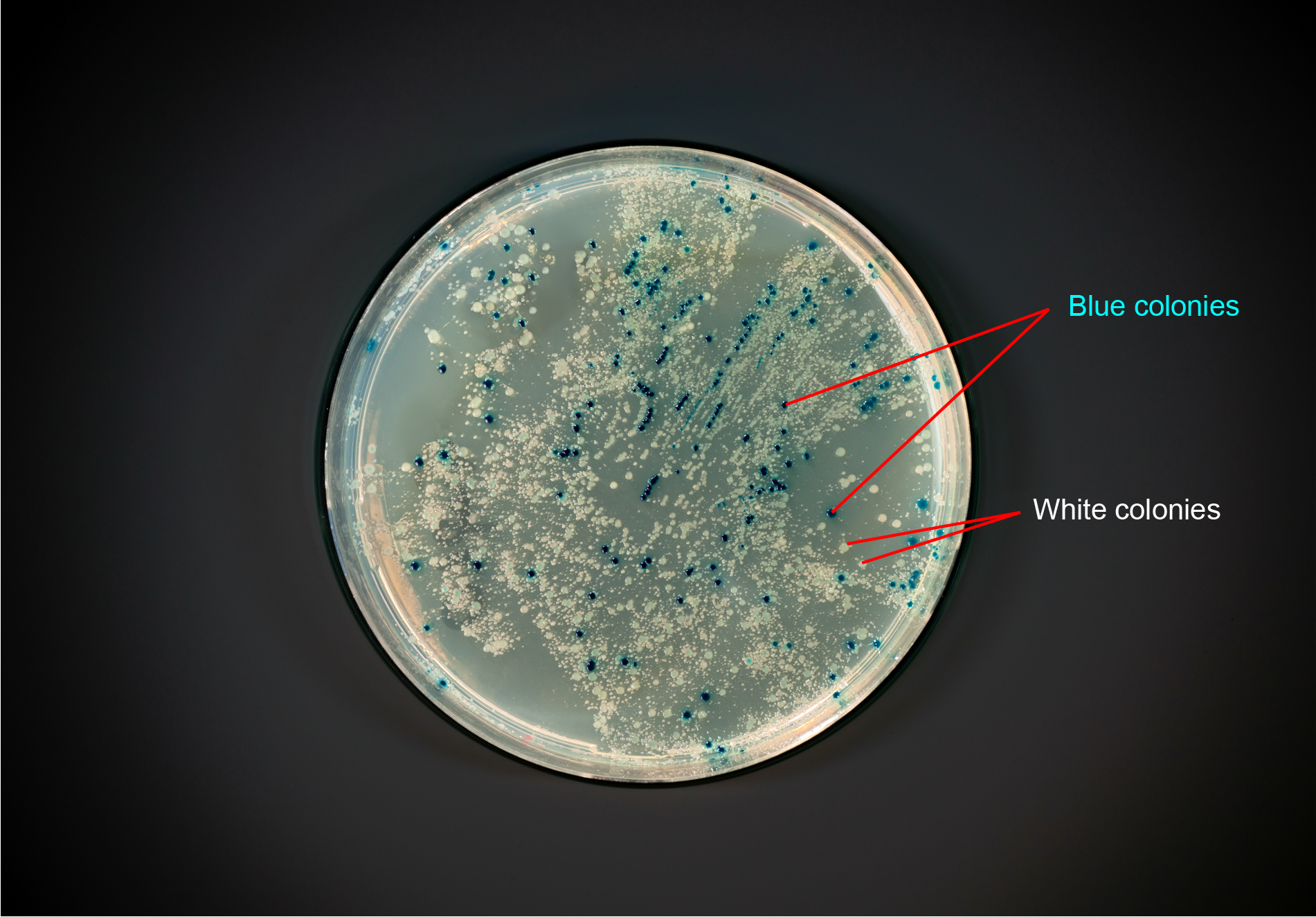
Figure 3. Photo of a plate with blue and white colonies.
In most cloning experiments, the colonies with the desired genetic construct are white, and the undesired colonies are blue.
This is achieved by a concept known as α-complementation (alpha-complementation) – a feature of most cloning host strains such as DH5-α.
Since this article is for illustrating how differential media is helpful during gene cloning, we will not go too much into the molecular genetics behind blue-white screening.
For that, we suggest looking at this article where we illustrate the concept of blue-white screening in detail.
Similarly, for experimental protocols related to blue-white screening, please refer to this document.
However, to put the concept of differential media into perspective, here is a brief description of what happens during blue-white screening.
The enzyme β-galactosidase is central to blue-white screening. β-galactosidase is synthesized by the lacZ gene as part of the lac operon, and β-galactosidase breaks down lactose.
Cloning host strains such as DH5-α encode a mutant form of the enzyme β-galactosidase, where amino acids 11-41 from the amino terminus are deleted. This mutant β-galactosidase is called ω-peptide that alone cannot breakdown lactose or other substrates of fully functional β-galactosidase enzymes such as X-gal.
Meanwhile, corresponding cloning vectors such as pUC19 encode a polypeptide called LacZ-α or α-peptide in the multiple cloning site. The LacZ-α / α-peptide makes up the 59 amino acids of the β-galactosidase enzyme.
When the ω-peptide is expressed by the host strain’s genome and the α-peptide is expressed by the vector, a fully functional β-galactosidase is formed that can breakdown lactose or its analogs. This is known as α-complementation.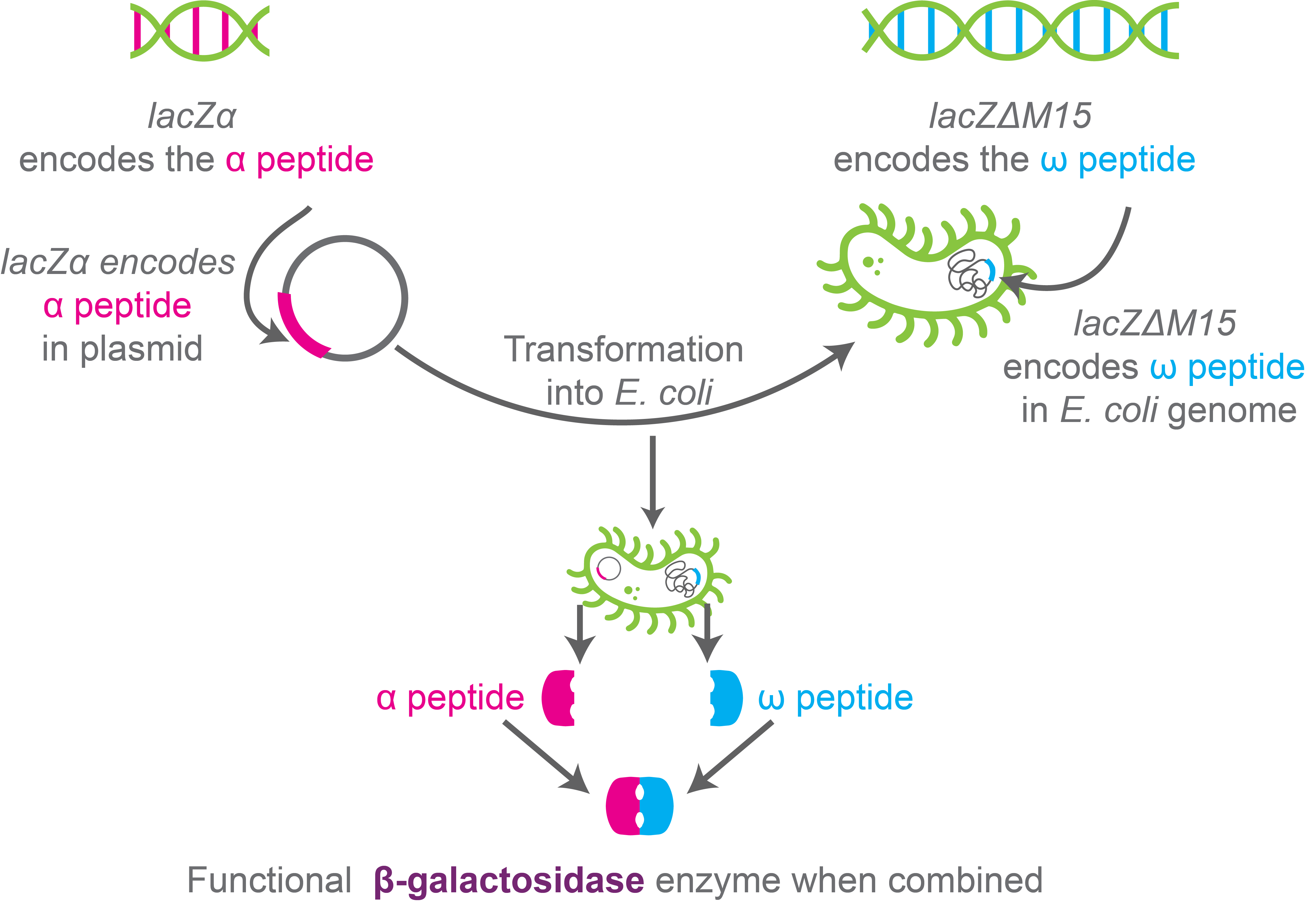
Figure 4. Schematic showing α-complementation.
So, when the host cells with the cloning vector are plated on media containing X-gal, the colonies that exhibit α-complementation produce functional β-galactosidase enzymes that break down X-gal, forming blue-colored products. The colonies, as a result, are blue.
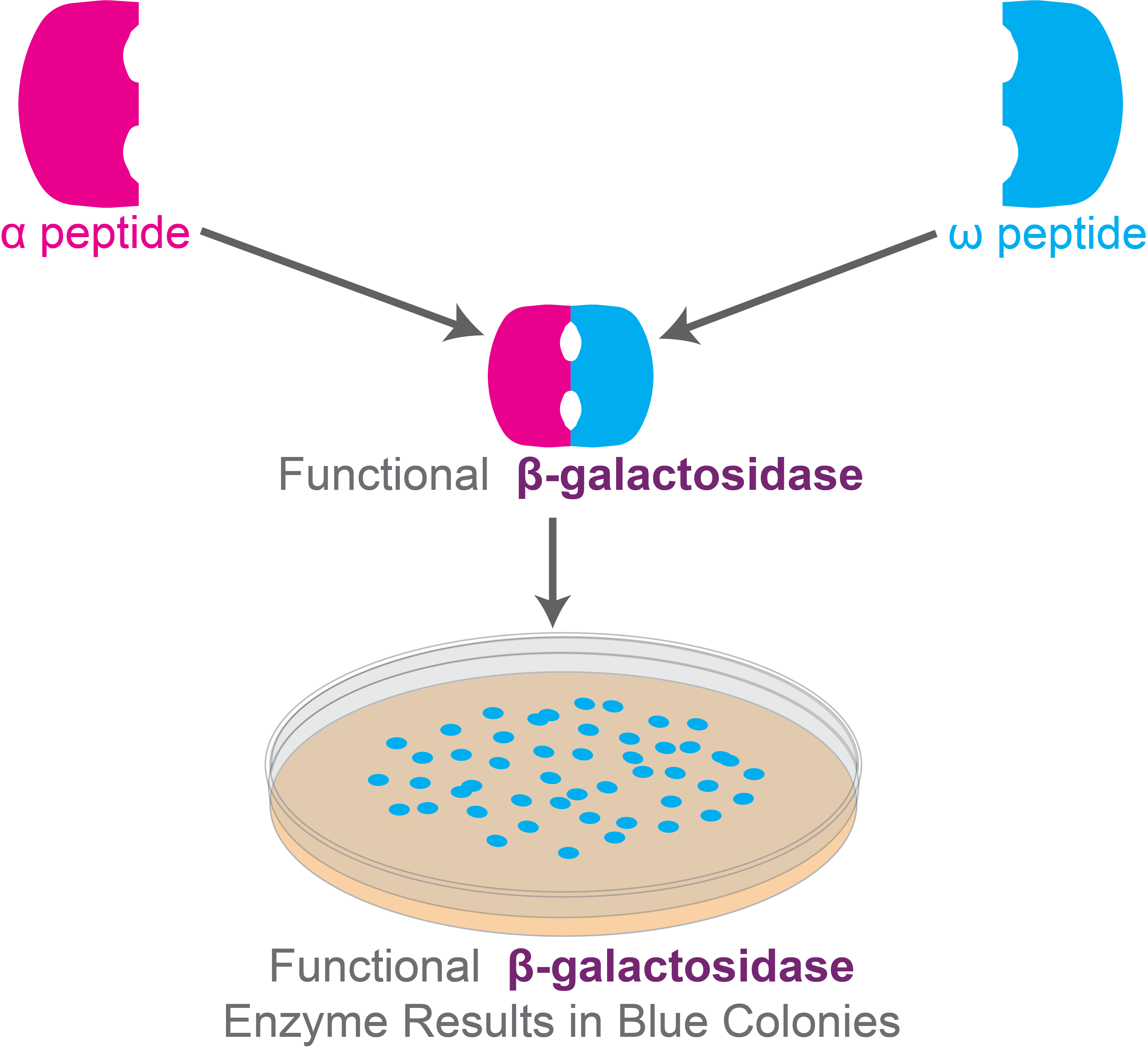 Figure 5. Functional β-galactosidase enzymes form blue colonies in the medium containing X-gal for blue-white screening experiment.
Figure 5. Functional β-galactosidase enzymes form blue colonies in the medium containing X-gal for blue-white screening experiment.
On the other hand, the host cells that do not have the α-peptide being expressed by the cloning vectors will not exhibit α-complementation, and no functional β-galactosidase enzymes are produced. Therefore, X-gal won’t be broken down by these cells. Their colonies will be white.
Please note that the gene for the α-peptide lies in the multiple cloning site (MCS) of the cloning vector. So, if the vector is blank – that is, no transgene is cloned within the MCS, then the α-peptide will still be produced. Here, α-complementation would occur, and the colony would be blue due to breakdown of X-gal by a functional β-galactosidase.
If the vector has a transgene cloned within its MCS the transgene would disrupt the gene for the α-peptide. The result of this disrupted gene is that there is no functional β-galactosidase enzyme produced, and X-gal won’t be broken down by these cells.

Figure 6. Disrupted lacZα sequence because of gene insertion does not encode α peptide.
And this is why the desired colonies on the plate will be white – the gene encoding the α-peptide was disrupted by the target transgene, which is what we want.
So, in this case, just by looking at the colonies you can easily tell which cells have the blank plasmid – they would be blue, and which colonies have the transgene insert – these colonies are white.

Figure 7. Schematic showing how to distinguish between colonies that have the plasmid with the inserted gene versus the ones that won’t in the blue-white screening experiment.
While this is the common way blue-white screening is utilized during genetic construct cloning, there is another approach for blue-white screening.
Here, the blue colonies represent the ones that have the necessary construct, and the white colonies do not have the construct.
This is when the genetic construct is cloned in the host strain’s genome rather than within a cloning vector the way we discussed above.
Look at the construct drawn in figure 8.
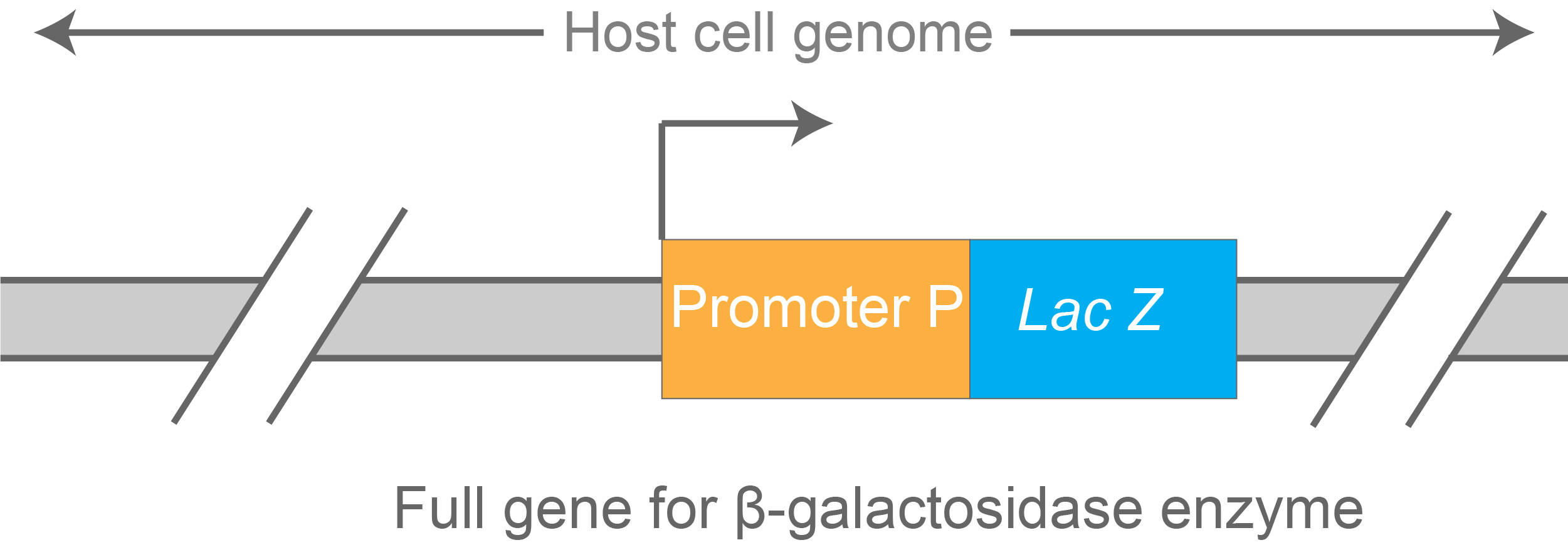
Figure 8. LacZ-based transcriptional reporter construct cloned in the host strain’s genome.
This construct depicts a reporter to measure transcription efficiency of promoter P. The stronger this promoter, the higher the level of lacZ transcription – and ultimately, levels of β- galactosidase enzyme synthesized by the cell.
Because of this, β- galactosidase enzyme activity can be used as a read-out for measuring the strength of this promoter.
These types of reporters are widely used in molecular biology.
Blue-white screening is employed during genetic engineering of such constructs. The colonies that show up as blue on an X-gal plate have the construct in their genomes. They are the desired colonies. The white colonies do not have the construct in their genomes; they are the undesired colonies.
Once the desired strain is made and screened via blue-white screening using differential media, the promoter strength is tested as the next step.
For this, the test strains and the control strains are grown up in liquid cultures to equal cell-densities, and then β- galactosidase enzyme activity is measured using Miller assay.
The result of this assay is used as a read-out for measuring the strength of the promoter to which lacZ is fused (as shown in figure 8 above). Higher the promoter strength is indicated by observing more corresponding β- galactosidase enzyme activity.
The thing to note here is that differential media and blue-white screening is used to screen the colonies that have the necessary genetic construct but not in the actual enzyme assay during which experimenters use a different substrate of β- galactosidase – such as ONPG(2-Nitrophenyl-β-D-galactopyranoside).Red-white screening is theoretically and technically identical to blue-white screening.
However, instead of X-gal, the substrate compound for the β- galactosidase enzyme is magenta-gal that is added to the medium. The product formed when magenta-gal is broken down by β- galactosidase is red.
So, depending on whether the colonies are producing a functional β-galactosidase or not, they would be red or white.
Very similar to blue-white screening, red-white screening is used during gene cloning to check which colonies have the necessary genetic construct cloned in them.

IPTG and auto-induction are two ways to induce protein expression in bacteria. They work similarly, but have different trade-offs in terms of convenience. While IPTG...

The final concentration of IPTG used for induction varies from 0.1 to 1.0 mM, with 0.5 or 1.0 mM most frequently used. For proteins with...

A His-tag is a stretch of 6-10 histidine amino acids in a row that is used for affinity purification, protein detection, and biochemical assays. His-tags...

Competent cells such as DH5a, DH10B, and BL21 will maintain their transformation efficiency for at least a year with proper storage. It is important to...
